Search any question & find its solution
Question:
Answered & Verified by Expert
$\mathrm{B}(\mathrm{OH})_3+\mathrm{NaOH} \rightleftharpoons \mathrm{NaBO}_2+\mathrm{Na}\left[\mathrm{B}(\mathrm{OH})_4\right]+\mathrm{H}_2 \mathrm{O}$.
How can this reaction is made to proceed in forward direction?
Options:
How can this reaction is made to proceed in forward direction?
Solution:
1254 Upvotes
Verified Answer
The correct answer is:
Addition of cis 1, 2 diol
Addition of cis 1, 2 diol
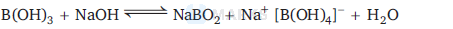
This reaction is reversible reaction because sodium metaborate, $\mathrm{Na}^{+}\left[\mathrm{B}(\mathrm{OH})_4\right]^{-}$formed by the reaction between $\mathrm{B}(\mathrm{OH})_3$ and $\mathrm{NaOH}$ gets hydrolysed to regenerate $\mathrm{B}(\mathrm{OH})_3$ and $\mathrm{NaOH}$
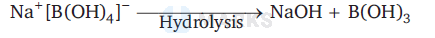
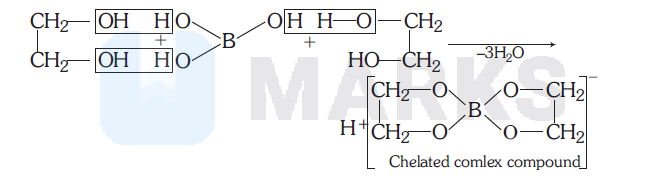
If, some quantity of polyhydroxy compound like cis-1, 3-diol, catechol, glycerol etc. is added to the reaction mixture. The $\mathrm{B}(\mathrm{OH})_3$ combines with such polyhydroxy compound to give chelated complex compound. Due to complex compound formation, stability increases and due to higher stability of complex, reaction moves in forward direction.
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.