Search any question & find its solution
Question:
Answered & Verified by Expert
Balance the following equations in basic medium by ion electron method and oxidation number methods and identify the oxidising agent and the reducing agent.
(a) \(\mathrm{P}_4(\mathrm{~s})+\mathrm{OH}^{-}\)(aq) \(\longrightarrow \mathrm{PH}_3(\mathrm{~g})+\mathrm{HPO}_2^{-}\)(aq)
(b) \(\mathrm{N}_2 \mathrm{H}_4(\ell)+\mathrm{ClO}_3^{-}(\mathrm{aq}) \longrightarrow \mathrm{NO}(\mathrm{g})+\mathrm{Cl}^{-}(\mathrm{g})\)
(c) \(\mathrm{Cl}_2 \mathrm{O}_7(\mathrm{~g})+\mathrm{H}_2 \mathrm{O}_2(\mathrm{aq}) \longrightarrow \mathrm{ClO}_2^{-}(\mathrm{aq})+\mathrm{O}_2(\mathrm{~g})+\mathrm{H}^{+}\)
(a) \(\mathrm{P}_4(\mathrm{~s})+\mathrm{OH}^{-}\)(aq) \(\longrightarrow \mathrm{PH}_3(\mathrm{~g})+\mathrm{HPO}_2^{-}\)(aq)
(b) \(\mathrm{N}_2 \mathrm{H}_4(\ell)+\mathrm{ClO}_3^{-}(\mathrm{aq}) \longrightarrow \mathrm{NO}(\mathrm{g})+\mathrm{Cl}^{-}(\mathrm{g})\)
(c) \(\mathrm{Cl}_2 \mathrm{O}_7(\mathrm{~g})+\mathrm{H}_2 \mathrm{O}_2(\mathrm{aq}) \longrightarrow \mathrm{ClO}_2^{-}(\mathrm{aq})+\mathrm{O}_2(\mathrm{~g})+\mathrm{H}^{+}\)
Solution:
2074 Upvotes
Verified Answer
(a) O. N. decreases by 3 per \(\mathrm{P}\) atom
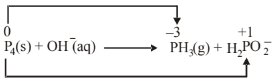
O. N. increases by 1 per P atom.
\(\mathrm{P}_4\) acts like both as an oxidizing as well as reducing agent.
Oxidation number method :
Total decrease in \(\mathrm{O}\). N. of \(\mathrm{P}_4\) in \(\mathrm{PH}_3=3 \times 4=12\)
Total increase in \(\mathrm{O}\). N. of \(\mathrm{P}_4\) in \(\mathrm{H}_2 \mathrm{PO}_2^{-}=1 \times 4=4\)
Therefore, to balance increase/decreases in \(\mathrm{O}\). N. multiply \(\mathrm{PH}_3\) by 1 and \(\mathrm{H}_2 \mathrm{PO}_2^{-}\)by 3 , we have,
\(\mathrm{P}_4(\mathrm{~s})+\mathrm{OH}^{-}(\mathrm{aq}) \longrightarrow \mathrm{PH}_3(\mathrm{~g})+3 \mathrm{H}_2 \mathrm{PO}_2^{-}(\mathrm{aq})\)
To balance \(\mathrm{O}\) atoms, multiply \(\mathrm{OH}^{-}\)by 6 , we have, \(\mathrm{P}_4(\mathrm{~s})+6 \mathrm{OH}^{-}(\mathrm{aq}) \longrightarrow \mathrm{PH}_3(\mathrm{~g})+3 \mathrm{H}_2 \mathrm{PO}_2^{-}(\mathrm{aq})\)
To balance \(\mathrm{H}\) atoms, add \(3 \mathrm{H}_2 \mathrm{O}\) to deficient side and \(3 \mathrm{OH}^{-}\) to the other side, now we have
\(\mathrm{P}_4(\mathrm{~s})+6 \mathrm{OH}^{-}(\mathrm{aq})+3 \mathrm{H}_2 \mathrm{O}(\ell) \longrightarrow\)
\(\mathrm{PH}_3(\mathrm{~g})+3 \mathrm{H}_2 \mathrm{PO}_2^{-}(\mathrm{aq})+3 \mathrm{OH}^{-}(\mathrm{aq})\)
To cancel out species present in both sides, we have \(\mathrm{P}_4(\mathrm{~s})+3 \mathrm{OH}^{-}(\mathrm{aq})+3 \mathrm{H}_2 \mathrm{O}(\ell) \longrightarrow\)
\(\mathrm{PH}_3(\mathrm{~g})+3 \mathrm{H}_2 \mathrm{PO}_2^{-}(\mathrm{aq})\)
This represents the correct balanced equation.
Ion electron method :
Oxidation half equation :
\(\stackrel{0}{\mathrm{P}_4} \longrightarrow \stackrel{+1}{\mathrm{H}_2 \mathrm{PO}_2^{-}}\)
Balance \(\mathrm{P}\) atoms, \(\mathrm{P}_4 \longrightarrow 4 \mathrm{H}_2 \mathrm{PO}_2^{-}\)
Balance \(\mathrm{H}\) atom \(\mathrm{O}\) atoms by adding \(\mathrm{OH}^{-}\)
\(\mathrm{P}_4+8 \mathrm{OH}^{-} \longrightarrow 4 \mathrm{H}_2 \mathrm{PO}_2{ }^{-}\)
Now balance charge
\(\mathrm{P}_4+8 \mathrm{OH}^{-} \longrightarrow 4 \mathrm{H}_2 \mathrm{PO}_2^{-}+4 \mathrm{e}^{-}\) ...(i)
Thus, Eq. (i) represents the correct balanced oxidation half equation.
Reduction half equation :
\(\stackrel{0}{\mathrm{P}_4} \longrightarrow \stackrel{-3}{\mathrm{PH}_3}\)
Balance \(\mathrm{P}\) atoms, \(\mathrm{P}_4 \longrightarrow 4 \mathrm{PH}_3\)
Balance \(\mathrm{H}\) atoms by adding \(\mathrm{OH}^{-}\)and \(\mathrm{H}_2 \mathrm{O}\), \(\mathrm{P}_4+12 \mathrm{H}_2 \mathrm{O}+12 \mathrm{e}^{-} \longrightarrow 4 \mathrm{PH}_3+12 \mathrm{OH}^{-} \ldots\) (ii)
Thus, Eq. (ii) represents the correct balanced reduction half equation.
To cancel out electrons gained and lost, multiply Eq. (i) by 3 and add with Eq. (ii) by cancelling common terms, we have,
\(\mathrm{P}_4(\mathrm{~s})+3 \mathrm{OH}^{-}(\mathrm{aq})+3 \mathrm{H}_2 \mathrm{O}(\ell) \longrightarrow\)
\(\mathrm{PH}_3(\mathrm{~g})+3 \mathrm{H}_2 \mathrm{PO}_2^{-}(\mathrm{aq})\)
(b)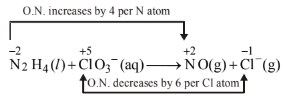
Thus, \(\mathrm{N}_2 \mathrm{H}_4\) is acting as a reducing agent while \(\mathrm{ClO}_3^{-}\)is acting as oxidising agent.
Oxidation number method :
Total increase in \(\mathrm{O}\). \(\mathrm{N}\). of \(\mathrm{N}\) in \(\mathrm{NO}=4 \times 2=8\)
Total increase in \(\mathrm{O} . \mathrm{N}\). of \(\mathrm{Cl}\) in
\(\mathrm{ClO}_3^{-}=6 \times 1=6\)
Therefore, to balance increase/decrease in O.N. multiply
\(\mathrm{NO}\) by 6 and \(\mathrm{ClO}_3{ }^{-}\)by 4 we have
\(\mathrm{N}_2 \mathrm{H}_4+4 \mathrm{ClO}_3^{-} \longrightarrow 6 \mathrm{NO}+\mathrm{Cl}^{-}\)
Balance \(\mathrm{N}\) and \(\mathrm{Cl}\) atoms,
\(3 \mathrm{~N}_2 \mathrm{H}_4+4 \mathrm{ClO}_3^{-} \longrightarrow 6 \mathrm{NO}+4 \mathrm{Cl}^{-}\)
To balance \(\mathrm{H}\) atoms, add \(6 \mathrm{H}_2 \mathrm{O}\) to deficient side
\(3 \mathrm{~N}_2 \mathrm{H}_4(\mathrm{~s})+4 \mathrm{ClO}_3^{-}(\mathrm{aq}) \longrightarrow 6 \mathrm{NO}(\mathrm{g})+4 \mathrm{Cl}^{-}(\mathrm{aq})+6 \mathrm{H}_2 \mathrm{O}(\ell)\)
This represents the correct balanced equation.
Ion electron method: Oxidation half equation:
\(\stackrel{-2}{\mathrm{N}_2} \mathrm{H}_4(\ell) \longrightarrow \stackrel{+2}{\mathrm{NO}}(\mathrm{g})\)
Balance \(\mathrm{N}\) atoms, \(\mathrm{N}_2 \mathrm{H}_4(\ell) \longrightarrow 2 \mathrm{NO}(\mathrm{g})\)
Balance \(\mathrm{H}\) atom by adding \(\mathrm{OH}^{-}\)and \(\mathrm{H}_2 \mathrm{O}\)
Balance \(\mathrm{O}\) atoms again by adding \(\mathrm{H}_2 \mathrm{O}\) and \(\mathrm{OH}^{-}\) \(\mathrm{N}_2 \mathrm{H}_4(\ell)+4 \mathrm{OH}^{-}(\mathrm{aq})+4 \mathrm{OH}^{-} \longrightarrow\)
\(2 \mathrm{NO}(\mathrm{g})+4 \mathrm{H}_2 \mathrm{O}(\ell)+2 \mathrm{H}_2 \mathrm{O}\)
Now balance charge
\(\mathrm{N}_2 \mathrm{H}_4(l)+8 \mathrm{OH}^{-}(\mathrm{aq}) \longrightarrow 2 \mathrm{NO}+6 \mathrm{H}_2 \mathrm{O}+8 \mathrm{e}^{-} \text {(i) }\) ...(i)
Thus, Eq. (i) represents the correct balanced oxidation half equation.
Reduction half equation \(\stackrel{+5}{\mathrm{Cl}}\mathrm{O}_3^{-}(\mathrm{aq}) \longrightarrow \mathrm{Cl}^{-}\)(aq)
Balance \(\mathrm{O}\). N. by adding electrons,
\(\mathrm{ClO}_3^{-}(\mathrm{aq})+6 \mathrm{e}^{-} \longrightarrow \mathrm{Cl}^{-}(\mathrm{aq})\)
Balance charge by adding \(\mathrm{OH}^{-}\)ions,
\(\mathrm{ClO}_3^{-}(\mathrm{aq})+6 \mathrm{e}^{-} \longrightarrow \mathrm{Cl}^{-}(\mathrm{aq})+6 \mathrm{OH}^{-}(\mathrm{aq})\)
Balance \(\mathrm{O}\) atoms by adding \(3 \mathrm{H}_2 \mathrm{O}\),
\(\mathrm{ClO}_3^{-}(\mathrm{aq})+3 \mathrm{H}_2 \mathrm{O}(\ell)+6 \mathrm{e}^{-} \longrightarrow\)
\(\mathrm{Cl}^{-}(\mathrm{aq})+6 \mathrm{OH}^{-}(\mathrm{aq}) \ldots \text { (ii) }\)
Thus, Eq. (ii) represents the correct balanced reduction half equation.
To cancel out electrons gained and lost, multiply Eq. (i) by 3 and Eq. (ii) by 4 and add, we have,
\(3 \mathrm{~N}_2 \mathrm{H}_4(\ell)+4 \mathrm{CIO}_3^{-}(\mathrm{aq}) \longrightarrow\)
\(6 \mathrm{NO}(\mathrm{g})+4 \mathrm{Cl}^{-}(\mathrm{aq})+6 \mathrm{H}_2 \mathrm{O}(\ell)\)
This represents the correct balanced equation
(c) O. N. decreases by 4 per \(\mathrm{Cl}\) atom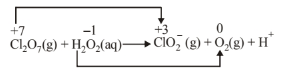
O. N. increases by 1 per \(\mathrm{O}\) atom. Thus, \(\mathrm{Cl}_2 \mathrm{O}_7(\mathrm{~g})\) acts an oxidizing agent while \(\mathrm{H}_2 \mathrm{O}_2\) (aq) as the reducing agent. Oxidation number method : Total decrease in \(\mathrm{O}\). N. of \(\mathrm{Cl}_2 \mathrm{O}_7=4 \times 2=8\)
Total increase in O. N. of \(\mathrm{H}_2 \mathrm{O}_2=2 \times 1=2\)
\(\therefore\) To balance increase/decrease in O. N. multiply \(\mathrm{H}_2 \mathrm{O}_2\) and \(\mathrm{O}_2\) by 4 , we have,
\(\mathrm{Cl}_2 \mathrm{O}_7(\mathrm{~g})+4 \mathrm{H}_2 \mathrm{O}_2(\mathrm{aq}) \longrightarrow \mathrm{ClO}_2^{-}(\mathrm{aq})+4 \mathrm{O}_2(\mathrm{~g})\)
To balance \(\mathrm{Cl}\) atoms, multiply \(\mathrm{ClO}_2^{-}\)by 2 , we have, \(\mathrm{Cl}_2 \mathrm{O}_7(\mathrm{~g})+4 \mathrm{H}_2 \mathrm{O}_2(\mathrm{aq}) \longrightarrow 2 \mathrm{ClO}_2^{-}(\mathrm{aq})+4 \mathrm{O}_2(\mathrm{~g})\)
To balance \(\mathrm{O}\) atoms, add \(3 \mathrm{H}_2 \mathrm{O}\) to R. H. S., we have, \(\mathrm{Cl}_2 \mathrm{O}_7(\mathrm{~g})+4 \mathrm{H}_2 \mathrm{O}_2(\mathrm{aq}) \longrightarrow\)
\(2 \mathrm{ClO}_2^{-}(\mathrm{aq})+4 \mathrm{O}_2(\mathrm{~g})+3 \mathrm{H}_2 \mathrm{O}(\ell)\)
To balance \(\mathrm{H}\) atoms, add \(2 \mathrm{H}_2 \mathrm{O}\) to R. H. S. and \(2 \mathrm{OH}^{-}\)to \(\mathrm{L}\).
H. S., we have,
\(\mathrm{Cl}_2 \mathrm{O}_7(\mathrm{~g})+4 \mathrm{H}_2 \mathrm{O}_2(\mathrm{~g})+2 \mathrm{OH}^{-}(\mathrm{aq}) \longrightarrow\)
\(2 \mathrm{ClO}_2^{-}(\mathrm{aq})+4 \mathrm{O}_2(\mathrm{~g})+3 \mathrm{H}_2 \mathrm{O}(\ell)\)
To balance \(\mathrm{H}\) atoms, add \(2 \mathrm{H}_2 \mathrm{O}\) to R. H. S. and \(2 \mathrm{OH}^{-}\)to \(\mathrm{L}\). H. S., we have,
\(\mathrm{Cl}_2 \mathrm{O}_7(\mathrm{~g})+4 \mathrm{H}_2 \mathrm{O}_2(\mathrm{~g})+2 \mathrm{OH}^{-}(\mathrm{aq}) \longrightarrow\)
\(2 \mathrm{ClO}_2^{-}(\mathrm{aq})+4 \mathrm{O}_2(\mathrm{~g})+5 \mathrm{H}_2 \mathrm{O}\)
This represents the balanced redox equation.
Ion electron method : Oxidation half equation:
Balance \(\mathrm{O}\). N. by adding electrons,
\(\mathrm{H}_2 \mathrm{O}_2(\mathrm{aq}) \longrightarrow \mathrm{O}_2(\mathrm{~g})+2 \mathrm{e}^{-}\)
Balance charge by adding \(\mathrm{OH}^{-}\)ions,
\(\mathrm{H}_2 \mathrm{O}_2(\mathrm{aq})+2 \mathrm{OH}^{-}(\mathrm{aq}) \longrightarrow \mathrm{O}_2(\mathrm{~g})+2 \mathrm{e}^{-}\)
Balance \(\mathrm{O}\) atoms by adding \(\mathrm{H}_2 \mathrm{O}\),
\(\mathrm{H}_2 \mathrm{O}_2(\mathrm{aq})+2 \mathrm{OH}^{-}(\mathrm{aq}) \longrightarrow \mathrm{O}_2(\mathrm{~g})+2 \mathrm{H}_2 \mathrm{O}(\ell)+2 \mathrm{e}^{-}\) ...(i)
Reduction half equation: \(\mathrm{Cl}_2 \mathrm{O}_7(\mathrm{~g}) \longrightarrow \mathrm{ClO}_2^{-}(\mathrm{aq})\)
Balance \(\mathrm{Cl}\) atoms; \(\mathrm{Cl}_2 \mathrm{O}_7(\mathrm{~g}) \longrightarrow 2 \mathrm{ClO}_2^{-}(\mathrm{aq})\)
Balance \(\mathrm{O}\). N. by adding electrons,
Add \(\mathrm{OH}^{-}\)ions to balance charge: \(\mathrm{Cl}_2 \mathrm{O}_7(\mathrm{~g})+8 \mathrm{e}^{-} \longrightarrow 2 \mathrm{ClO}_2^{-}(\mathrm{aq})+6 \mathrm{OH}^{-}\)
Balance \(\mathrm{O}\) atoms by adding \(3 \mathrm{H}_2 \mathrm{O}\) to \(\mathrm{L}\). H. S., we have,
\(\mathrm{Cl}_2 \mathrm{O}_7(\mathrm{~g})+3 \mathrm{H}_2 \mathrm{O}(\ell)+8 \mathrm{e}^{-} \longrightarrow\)
\(2 \mathrm{ClO}_2^{-}(\mathrm{aq})+6 \mathrm{OH}^{-}(\mathrm{aq}) \quad \ldots\) (ii)
To cancel out electrons, multiply Eq. (i) by 4 and add it to Eq. (ii), we have,
\(4 \mathrm{H}_2 \mathrm{O}_2(\mathrm{aq})+8 \mathrm{OH}^{-}(\mathrm{aq})+\mathrm{Cl}_2 \mathrm{O}_7(\mathrm{~g})+3 \mathrm{H}_2 \mathrm{O}(\ell) \longrightarrow 2\)
\(\mathrm{ClO}_2^{-}+(\mathrm{aq})+6 \mathrm{OH}^{-}(\mathrm{aq})+4 \mathrm{O}_2(\mathrm{~g})+8 \mathrm{H}_2 \mathrm{O}(\ell)\)
or \(\mathrm{Cl}_2 \mathrm{O}_7(\mathrm{~g})+4 \mathrm{H}_2 \mathrm{O}_2(\mathrm{aq})+2 \mathrm{OH}^{-}(\mathrm{aq}) \longrightarrow 2 \mathrm{ClO}_2^{-}\)
\((\mathrm{aq})+4 \mathrm{O}_2(\mathrm{~g})+5 \mathrm{H}_2 \mathrm{O}(\ell)\)

O. N. increases by 1 per P atom.
\(\mathrm{P}_4\) acts like both as an oxidizing as well as reducing agent.
Oxidation number method :
Total decrease in \(\mathrm{O}\). N. of \(\mathrm{P}_4\) in \(\mathrm{PH}_3=3 \times 4=12\)
Total increase in \(\mathrm{O}\). N. of \(\mathrm{P}_4\) in \(\mathrm{H}_2 \mathrm{PO}_2^{-}=1 \times 4=4\)
Therefore, to balance increase/decreases in \(\mathrm{O}\). N. multiply \(\mathrm{PH}_3\) by 1 and \(\mathrm{H}_2 \mathrm{PO}_2^{-}\)by 3 , we have,
\(\mathrm{P}_4(\mathrm{~s})+\mathrm{OH}^{-}(\mathrm{aq}) \longrightarrow \mathrm{PH}_3(\mathrm{~g})+3 \mathrm{H}_2 \mathrm{PO}_2^{-}(\mathrm{aq})\)
To balance \(\mathrm{O}\) atoms, multiply \(\mathrm{OH}^{-}\)by 6 , we have, \(\mathrm{P}_4(\mathrm{~s})+6 \mathrm{OH}^{-}(\mathrm{aq}) \longrightarrow \mathrm{PH}_3(\mathrm{~g})+3 \mathrm{H}_2 \mathrm{PO}_2^{-}(\mathrm{aq})\)
To balance \(\mathrm{H}\) atoms, add \(3 \mathrm{H}_2 \mathrm{O}\) to deficient side and \(3 \mathrm{OH}^{-}\) to the other side, now we have
\(\mathrm{P}_4(\mathrm{~s})+6 \mathrm{OH}^{-}(\mathrm{aq})+3 \mathrm{H}_2 \mathrm{O}(\ell) \longrightarrow\)
\(\mathrm{PH}_3(\mathrm{~g})+3 \mathrm{H}_2 \mathrm{PO}_2^{-}(\mathrm{aq})+3 \mathrm{OH}^{-}(\mathrm{aq})\)
To cancel out species present in both sides, we have \(\mathrm{P}_4(\mathrm{~s})+3 \mathrm{OH}^{-}(\mathrm{aq})+3 \mathrm{H}_2 \mathrm{O}(\ell) \longrightarrow\)
\(\mathrm{PH}_3(\mathrm{~g})+3 \mathrm{H}_2 \mathrm{PO}_2^{-}(\mathrm{aq})\)
This represents the correct balanced equation.
Ion electron method :
Oxidation half equation :
\(\stackrel{0}{\mathrm{P}_4} \longrightarrow \stackrel{+1}{\mathrm{H}_2 \mathrm{PO}_2^{-}}\)
Balance \(\mathrm{P}\) atoms, \(\mathrm{P}_4 \longrightarrow 4 \mathrm{H}_2 \mathrm{PO}_2^{-}\)
Balance \(\mathrm{H}\) atom \(\mathrm{O}\) atoms by adding \(\mathrm{OH}^{-}\)
\(\mathrm{P}_4+8 \mathrm{OH}^{-} \longrightarrow 4 \mathrm{H}_2 \mathrm{PO}_2{ }^{-}\)
Now balance charge
\(\mathrm{P}_4+8 \mathrm{OH}^{-} \longrightarrow 4 \mathrm{H}_2 \mathrm{PO}_2^{-}+4 \mathrm{e}^{-}\) ...(i)
Thus, Eq. (i) represents the correct balanced oxidation half equation.
Reduction half equation :
\(\stackrel{0}{\mathrm{P}_4} \longrightarrow \stackrel{-3}{\mathrm{PH}_3}\)
Balance \(\mathrm{P}\) atoms, \(\mathrm{P}_4 \longrightarrow 4 \mathrm{PH}_3\)
Balance \(\mathrm{H}\) atoms by adding \(\mathrm{OH}^{-}\)and \(\mathrm{H}_2 \mathrm{O}\), \(\mathrm{P}_4+12 \mathrm{H}_2 \mathrm{O}+12 \mathrm{e}^{-} \longrightarrow 4 \mathrm{PH}_3+12 \mathrm{OH}^{-} \ldots\) (ii)
Thus, Eq. (ii) represents the correct balanced reduction half equation.
To cancel out electrons gained and lost, multiply Eq. (i) by 3 and add with Eq. (ii) by cancelling common terms, we have,
\(\mathrm{P}_4(\mathrm{~s})+3 \mathrm{OH}^{-}(\mathrm{aq})+3 \mathrm{H}_2 \mathrm{O}(\ell) \longrightarrow\)
\(\mathrm{PH}_3(\mathrm{~g})+3 \mathrm{H}_2 \mathrm{PO}_2^{-}(\mathrm{aq})\)
(b)

Thus, \(\mathrm{N}_2 \mathrm{H}_4\) is acting as a reducing agent while \(\mathrm{ClO}_3^{-}\)is acting as oxidising agent.
Oxidation number method :
Total increase in \(\mathrm{O}\). \(\mathrm{N}\). of \(\mathrm{N}\) in \(\mathrm{NO}=4 \times 2=8\)
Total increase in \(\mathrm{O} . \mathrm{N}\). of \(\mathrm{Cl}\) in
\(\mathrm{ClO}_3^{-}=6 \times 1=6\)
Therefore, to balance increase/decrease in O.N. multiply
\(\mathrm{NO}\) by 6 and \(\mathrm{ClO}_3{ }^{-}\)by 4 we have
\(\mathrm{N}_2 \mathrm{H}_4+4 \mathrm{ClO}_3^{-} \longrightarrow 6 \mathrm{NO}+\mathrm{Cl}^{-}\)
Balance \(\mathrm{N}\) and \(\mathrm{Cl}\) atoms,
\(3 \mathrm{~N}_2 \mathrm{H}_4+4 \mathrm{ClO}_3^{-} \longrightarrow 6 \mathrm{NO}+4 \mathrm{Cl}^{-}\)
To balance \(\mathrm{H}\) atoms, add \(6 \mathrm{H}_2 \mathrm{O}\) to deficient side
\(3 \mathrm{~N}_2 \mathrm{H}_4(\mathrm{~s})+4 \mathrm{ClO}_3^{-}(\mathrm{aq}) \longrightarrow 6 \mathrm{NO}(\mathrm{g})+4 \mathrm{Cl}^{-}(\mathrm{aq})+6 \mathrm{H}_2 \mathrm{O}(\ell)\)
This represents the correct balanced equation.
Ion electron method: Oxidation half equation:
\(\stackrel{-2}{\mathrm{N}_2} \mathrm{H}_4(\ell) \longrightarrow \stackrel{+2}{\mathrm{NO}}(\mathrm{g})\)
Balance \(\mathrm{N}\) atoms, \(\mathrm{N}_2 \mathrm{H}_4(\ell) \longrightarrow 2 \mathrm{NO}(\mathrm{g})\)
Balance \(\mathrm{H}\) atom by adding \(\mathrm{OH}^{-}\)and \(\mathrm{H}_2 \mathrm{O}\)
Balance \(\mathrm{O}\) atoms again by adding \(\mathrm{H}_2 \mathrm{O}\) and \(\mathrm{OH}^{-}\) \(\mathrm{N}_2 \mathrm{H}_4(\ell)+4 \mathrm{OH}^{-}(\mathrm{aq})+4 \mathrm{OH}^{-} \longrightarrow\)
\(2 \mathrm{NO}(\mathrm{g})+4 \mathrm{H}_2 \mathrm{O}(\ell)+2 \mathrm{H}_2 \mathrm{O}\)
Now balance charge
\(\mathrm{N}_2 \mathrm{H}_4(l)+8 \mathrm{OH}^{-}(\mathrm{aq}) \longrightarrow 2 \mathrm{NO}+6 \mathrm{H}_2 \mathrm{O}+8 \mathrm{e}^{-} \text {(i) }\) ...(i)
Thus, Eq. (i) represents the correct balanced oxidation half equation.
Reduction half equation \(\stackrel{+5}{\mathrm{Cl}}\mathrm{O}_3^{-}(\mathrm{aq}) \longrightarrow \mathrm{Cl}^{-}\)(aq)
Balance \(\mathrm{O}\). N. by adding electrons,
\(\mathrm{ClO}_3^{-}(\mathrm{aq})+6 \mathrm{e}^{-} \longrightarrow \mathrm{Cl}^{-}(\mathrm{aq})\)
Balance charge by adding \(\mathrm{OH}^{-}\)ions,
\(\mathrm{ClO}_3^{-}(\mathrm{aq})+6 \mathrm{e}^{-} \longrightarrow \mathrm{Cl}^{-}(\mathrm{aq})+6 \mathrm{OH}^{-}(\mathrm{aq})\)
Balance \(\mathrm{O}\) atoms by adding \(3 \mathrm{H}_2 \mathrm{O}\),
\(\mathrm{ClO}_3^{-}(\mathrm{aq})+3 \mathrm{H}_2 \mathrm{O}(\ell)+6 \mathrm{e}^{-} \longrightarrow\)
\(\mathrm{Cl}^{-}(\mathrm{aq})+6 \mathrm{OH}^{-}(\mathrm{aq}) \ldots \text { (ii) }\)
Thus, Eq. (ii) represents the correct balanced reduction half equation.
To cancel out electrons gained and lost, multiply Eq. (i) by 3 and Eq. (ii) by 4 and add, we have,
\(3 \mathrm{~N}_2 \mathrm{H}_4(\ell)+4 \mathrm{CIO}_3^{-}(\mathrm{aq}) \longrightarrow\)
\(6 \mathrm{NO}(\mathrm{g})+4 \mathrm{Cl}^{-}(\mathrm{aq})+6 \mathrm{H}_2 \mathrm{O}(\ell)\)
This represents the correct balanced equation
(c) O. N. decreases by 4 per \(\mathrm{Cl}\) atom

O. N. increases by 1 per \(\mathrm{O}\) atom. Thus, \(\mathrm{Cl}_2 \mathrm{O}_7(\mathrm{~g})\) acts an oxidizing agent while \(\mathrm{H}_2 \mathrm{O}_2\) (aq) as the reducing agent. Oxidation number method : Total decrease in \(\mathrm{O}\). N. of \(\mathrm{Cl}_2 \mathrm{O}_7=4 \times 2=8\)
Total increase in O. N. of \(\mathrm{H}_2 \mathrm{O}_2=2 \times 1=2\)
\(\therefore\) To balance increase/decrease in O. N. multiply \(\mathrm{H}_2 \mathrm{O}_2\) and \(\mathrm{O}_2\) by 4 , we have,
\(\mathrm{Cl}_2 \mathrm{O}_7(\mathrm{~g})+4 \mathrm{H}_2 \mathrm{O}_2(\mathrm{aq}) \longrightarrow \mathrm{ClO}_2^{-}(\mathrm{aq})+4 \mathrm{O}_2(\mathrm{~g})\)
To balance \(\mathrm{Cl}\) atoms, multiply \(\mathrm{ClO}_2^{-}\)by 2 , we have, \(\mathrm{Cl}_2 \mathrm{O}_7(\mathrm{~g})+4 \mathrm{H}_2 \mathrm{O}_2(\mathrm{aq}) \longrightarrow 2 \mathrm{ClO}_2^{-}(\mathrm{aq})+4 \mathrm{O}_2(\mathrm{~g})\)
To balance \(\mathrm{O}\) atoms, add \(3 \mathrm{H}_2 \mathrm{O}\) to R. H. S., we have, \(\mathrm{Cl}_2 \mathrm{O}_7(\mathrm{~g})+4 \mathrm{H}_2 \mathrm{O}_2(\mathrm{aq}) \longrightarrow\)
\(2 \mathrm{ClO}_2^{-}(\mathrm{aq})+4 \mathrm{O}_2(\mathrm{~g})+3 \mathrm{H}_2 \mathrm{O}(\ell)\)
To balance \(\mathrm{H}\) atoms, add \(2 \mathrm{H}_2 \mathrm{O}\) to R. H. S. and \(2 \mathrm{OH}^{-}\)to \(\mathrm{L}\).
H. S., we have,
\(\mathrm{Cl}_2 \mathrm{O}_7(\mathrm{~g})+4 \mathrm{H}_2 \mathrm{O}_2(\mathrm{~g})+2 \mathrm{OH}^{-}(\mathrm{aq}) \longrightarrow\)
\(2 \mathrm{ClO}_2^{-}(\mathrm{aq})+4 \mathrm{O}_2(\mathrm{~g})+3 \mathrm{H}_2 \mathrm{O}(\ell)\)
To balance \(\mathrm{H}\) atoms, add \(2 \mathrm{H}_2 \mathrm{O}\) to R. H. S. and \(2 \mathrm{OH}^{-}\)to \(\mathrm{L}\). H. S., we have,
\(\mathrm{Cl}_2 \mathrm{O}_7(\mathrm{~g})+4 \mathrm{H}_2 \mathrm{O}_2(\mathrm{~g})+2 \mathrm{OH}^{-}(\mathrm{aq}) \longrightarrow\)
\(2 \mathrm{ClO}_2^{-}(\mathrm{aq})+4 \mathrm{O}_2(\mathrm{~g})+5 \mathrm{H}_2 \mathrm{O}\)
This represents the balanced redox equation.
Ion electron method : Oxidation half equation:
Balance \(\mathrm{O}\). N. by adding electrons,
\(\mathrm{H}_2 \mathrm{O}_2(\mathrm{aq}) \longrightarrow \mathrm{O}_2(\mathrm{~g})+2 \mathrm{e}^{-}\)
Balance charge by adding \(\mathrm{OH}^{-}\)ions,
\(\mathrm{H}_2 \mathrm{O}_2(\mathrm{aq})+2 \mathrm{OH}^{-}(\mathrm{aq}) \longrightarrow \mathrm{O}_2(\mathrm{~g})+2 \mathrm{e}^{-}\)
Balance \(\mathrm{O}\) atoms by adding \(\mathrm{H}_2 \mathrm{O}\),
\(\mathrm{H}_2 \mathrm{O}_2(\mathrm{aq})+2 \mathrm{OH}^{-}(\mathrm{aq}) \longrightarrow \mathrm{O}_2(\mathrm{~g})+2 \mathrm{H}_2 \mathrm{O}(\ell)+2 \mathrm{e}^{-}\) ...(i)
Reduction half equation: \(\mathrm{Cl}_2 \mathrm{O}_7(\mathrm{~g}) \longrightarrow \mathrm{ClO}_2^{-}(\mathrm{aq})\)
Balance \(\mathrm{Cl}\) atoms; \(\mathrm{Cl}_2 \mathrm{O}_7(\mathrm{~g}) \longrightarrow 2 \mathrm{ClO}_2^{-}(\mathrm{aq})\)
Balance \(\mathrm{O}\). N. by adding electrons,
Add \(\mathrm{OH}^{-}\)ions to balance charge: \(\mathrm{Cl}_2 \mathrm{O}_7(\mathrm{~g})+8 \mathrm{e}^{-} \longrightarrow 2 \mathrm{ClO}_2^{-}(\mathrm{aq})+6 \mathrm{OH}^{-}\)
Balance \(\mathrm{O}\) atoms by adding \(3 \mathrm{H}_2 \mathrm{O}\) to \(\mathrm{L}\). H. S., we have,
\(\mathrm{Cl}_2 \mathrm{O}_7(\mathrm{~g})+3 \mathrm{H}_2 \mathrm{O}(\ell)+8 \mathrm{e}^{-} \longrightarrow\)
\(2 \mathrm{ClO}_2^{-}(\mathrm{aq})+6 \mathrm{OH}^{-}(\mathrm{aq}) \quad \ldots\) (ii)
To cancel out electrons, multiply Eq. (i) by 4 and add it to Eq. (ii), we have,
\(4 \mathrm{H}_2 \mathrm{O}_2(\mathrm{aq})+8 \mathrm{OH}^{-}(\mathrm{aq})+\mathrm{Cl}_2 \mathrm{O}_7(\mathrm{~g})+3 \mathrm{H}_2 \mathrm{O}(\ell) \longrightarrow 2\)
\(\mathrm{ClO}_2^{-}+(\mathrm{aq})+6 \mathrm{OH}^{-}(\mathrm{aq})+4 \mathrm{O}_2(\mathrm{~g})+8 \mathrm{H}_2 \mathrm{O}(\ell)\)
or \(\mathrm{Cl}_2 \mathrm{O}_7(\mathrm{~g})+4 \mathrm{H}_2 \mathrm{O}_2(\mathrm{aq})+2 \mathrm{OH}^{-}(\mathrm{aq}) \longrightarrow 2 \mathrm{ClO}_2^{-}\)
\((\mathrm{aq})+4 \mathrm{O}_2(\mathrm{~g})+5 \mathrm{H}_2 \mathrm{O}(\ell)\)
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.