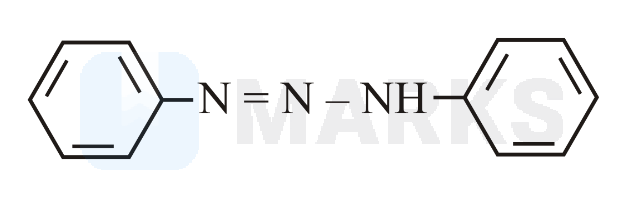Search any question & find its solution
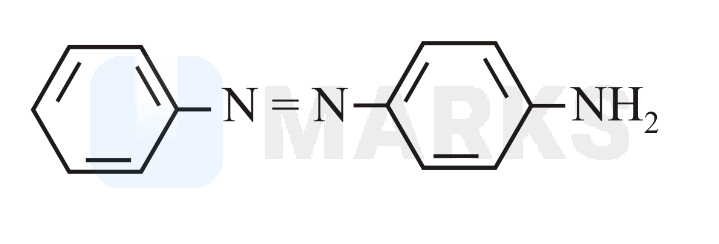
Aniline reacts with a mixture of sodium nitrite and hydrochloric acid and forms benzene diazonium chloride.The structure of Benzene diazonium chloride is

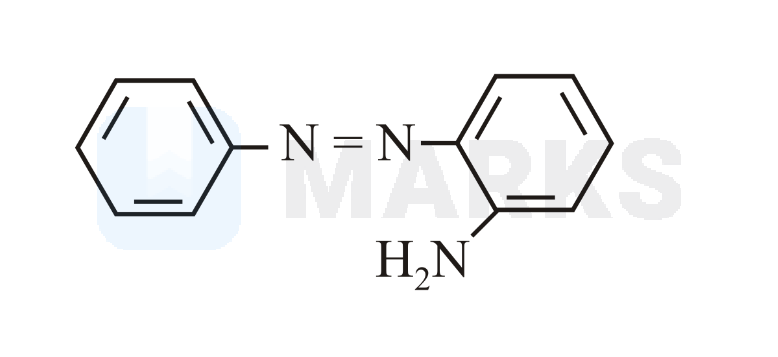
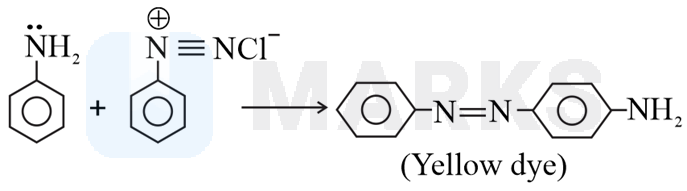
Benzene diazonium chloride reacts with aniline in presence of dilute hydrochloric acid and undergoes coupling reaction and produces stable azo products. In this reaction, benzene diazonium chloride acts as an electrophile in coupling with activated aniline. In this type of reaction, substitution takes place at the para position. So, the reaction Benzene diazonium chloride with aniline in presence of dilute hydrochloric acid is as follows:
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.