Search any question & find its solution
Question:
Answered & Verified by Expert
Carnot cycle of an engine is given below
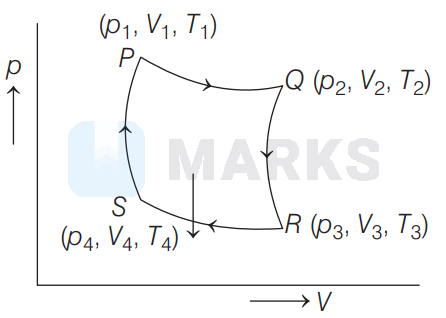
Total work done by the gas in one cycle is
Options:

Total work done by the gas in one cycle is
Solution:
2395 Upvotes
Verified Answer
The correct answer is:
$\mu R T_{1} \log \frac{V_{2}}{V_{1}}-\mu R T_{2} \log \frac{V_{3}}{V_{4}}$
Total work done by the gas
$$
W_{\text {Total }}=W_{A \rightarrow B}+W_{B \rightarrow C}+W_{C \rightarrow D}+W_{D \rightarrow A}
$$
Work done in isothermal process by ideal gas
$$
W_{A \rightarrow B}=\mu R T, \log \frac{V_{2}}{V_{1}}
$$
Work done in adiabatic process by ideal gas
$$
W_{B \rightarrow C}=\frac{\mu R\left(T_{1}-T_{2}\right)}{r-1}
$$
Work done in isothermal process by ideal gas,
$$
W_{C \rightarrow D}=-\mu R T_{2} \log \frac{V_{3}}{V_{4}}
$$
Work done in adiabatic process, by ideal gas
$$
W_{D \rightarrow A}=\frac{-\mu R\left(T_{1}-T_{2}\right)}{r-1}
$$
Adding Eqs. (i), (ii), (iii) and (iv) we get
$$
W_{\text {Total }}=\mu R T_{1} \log \frac{V_{2}}{V_{1}}-\mu R T_{2} \log \frac{V_{3}}{V_{4}}
$$
$$
W_{\text {Total }}=W_{A \rightarrow B}+W_{B \rightarrow C}+W_{C \rightarrow D}+W_{D \rightarrow A}
$$
Work done in isothermal process by ideal gas
$$
W_{A \rightarrow B}=\mu R T, \log \frac{V_{2}}{V_{1}}
$$
Work done in adiabatic process by ideal gas
$$
W_{B \rightarrow C}=\frac{\mu R\left(T_{1}-T_{2}\right)}{r-1}
$$
Work done in isothermal process by ideal gas,
$$
W_{C \rightarrow D}=-\mu R T_{2} \log \frac{V_{3}}{V_{4}}
$$
Work done in adiabatic process, by ideal gas
$$
W_{D \rightarrow A}=\frac{-\mu R\left(T_{1}-T_{2}\right)}{r-1}
$$
Adding Eqs. (i), (ii), (iii) and (iv) we get
$$
W_{\text {Total }}=\mu R T_{1} \log \frac{V_{2}}{V_{1}}-\mu R T_{2} \log \frac{V_{3}}{V_{4}}
$$
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.