Search any question & find its solution
Question:
Answered & Verified by Expert
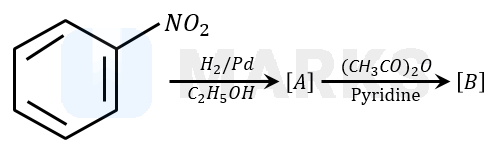
Solution:
2660 Upvotes
Verified Answer
The correct answer is:
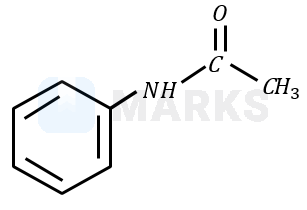

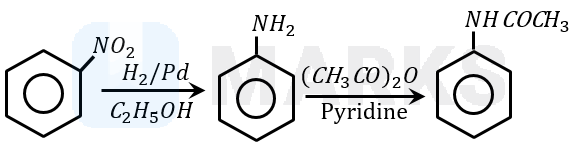
Nitrobenzene is reduced to aniline by passing hydrogen gas in presence of finely divided palladium and also by reduction with metals in an acidic medium. When aniline reacts with acetic anhydride in the presence of pyridine to give acetanilide. Aniline acts as a weak base, while acetic anhydride acts as electrophile.
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.