Search any question & find its solution
Chlorine undergoes disproportionation in alkaline medium as shown below :
The values of and in a balanced redox reaction are respectively :
Chlorine undergoes disproportionation reaction in alkaline medium. It is the simultaneous oxidation-reduction. Chlorine is simultaneously reduced to chloride ion and is oxidised to ion. Halogens (e.g. Cl) have a strong tendency to accept electrons, so they act as strong oxidising agents.
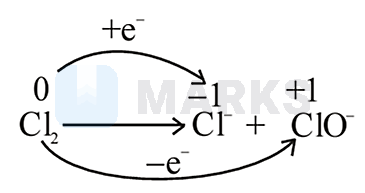
The reaction is as follows,
\(\Rightarrow \mathrm{Cl}_2 - 2 \mathrm{O} \mathrm{H}^- \longrightarrow \mathrm{Cl}^{-}+\mathrm{ClO}^{-}+\mathrm{H}_2 \mathrm{O}\)
Hence the values of a,b,c,d are
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.