Search any question & find its solution
Question:
Answered & Verified by Expert
Compounds ' $\mathrm{A}^{\prime}$ with molecular formula $\mathrm{C}_4 \mathrm{H}_9$ Br is treated with aq. $\mathrm{KOH}$ solution. The rate of this reaction depends upon the concentration of the compound 'A' only. When another optically active isomer ' $\mathrm{B}$ ' of this compound was treated with aq. $\mathrm{KOH}$ solution, the rate of reaction was found to be dependent on concentration of compound and $\mathrm{KOH}$ both.
(i) Write down the structural formula of both compounds 'A' and 'B'.
(ii) Out of these two compounds, which one will be converted to the product with inverted configuration.
(i) Write down the structural formula of both compounds 'A' and 'B'.
(ii) Out of these two compounds, which one will be converted to the product with inverted configuration.
Solution:
1105 Upvotes
Verified Answer
(i) Since the rate of reaction depends upon the concentration of compound 'A' $\left(\mathrm{C}_4 \mathrm{H}_9 \mathrm{Br}\right)$ only therefore, the reaction follows $\mathrm{S}_{\mathrm{N}^1}$ mechanism and the given compound will be 2-bromo-2methylpropane i.e. tertiary alkyl halide and its structure is as follows:
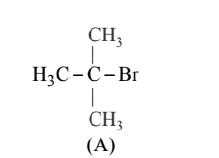
2-bromobutane (B) is the optically active isomer of (A) and its structural formula is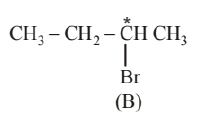
(ii) Compound (B) will be converted to the product with inverted configuration. The rate of reaction of compound (B) depends both upon the concentration of compound (B) and $\mathrm{KOH}$, So, the reaction follow $\mathrm{S}_{\mathrm{N}} 2$ mechanism. In $\mathrm{S}_{\mathrm{N}} 2$ reaction, attack of the nucleophile is from the back side, therefore, the product of hydrolysis will have inverted configuration.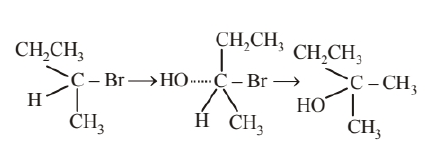

2-bromobutane (B) is the optically active isomer of (A) and its structural formula is
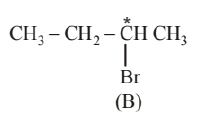
(ii) Compound (B) will be converted to the product with inverted configuration. The rate of reaction of compound (B) depends both upon the concentration of compound (B) and $\mathrm{KOH}$, So, the reaction follow $\mathrm{S}_{\mathrm{N}} 2$ mechanism. In $\mathrm{S}_{\mathrm{N}} 2$ reaction, attack of the nucleophile is from the back side, therefore, the product of hydrolysis will have inverted configuration.

Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.