Search any question & find its solution
Question:
Answered & Verified by Expert
Consider the Born-Haber cycle for the formation of an ionic compound given below and identify the compound ( ) formed.
$$
\left[\begin{array}{l}
\mathrm{M}(\mathrm{s}) \stackrel{\Delta \mathrm{H}_{1}}{\longrightarrow} \mathrm{M}(\mathrm{g}) \stackrel{\Delta \mathrm{H}_{2}}{\longrightarrow} \mathrm{M}^{+}(\mathrm{g}) \\
\frac{1}{2} \mathrm{X}_{2}(\mathrm{~g}) \stackrel{\Delta \mathrm{H}_{3}}{\longrightarrow} \mathrm{X}(\mathrm{g}) \stackrel{\Delta \mathrm{H}_{4}}{\longrightarrow} \mathrm{X}^{-}(\mathrm{g})
\end{array}\right] \stackrel{\Delta \mathrm{H}_{5}}{\longrightarrow} \mathrm{Z}
$$
Options:
$$
\left[\begin{array}{l}
\mathrm{M}(\mathrm{s}) \stackrel{\Delta \mathrm{H}_{1}}{\longrightarrow} \mathrm{M}(\mathrm{g}) \stackrel{\Delta \mathrm{H}_{2}}{\longrightarrow} \mathrm{M}^{+}(\mathrm{g}) \\
\frac{1}{2} \mathrm{X}_{2}(\mathrm{~g}) \stackrel{\Delta \mathrm{H}_{3}}{\longrightarrow} \mathrm{X}(\mathrm{g}) \stackrel{\Delta \mathrm{H}_{4}}{\longrightarrow} \mathrm{X}^{-}(\mathrm{g})
\end{array}\right] \stackrel{\Delta \mathrm{H}_{5}}{\longrightarrow} \mathrm{Z}
$$
Solution:
2746 Upvotes
Verified Answer
The correct answer is:
$\mathrm{M}^{+} \mathrm{X}^{-}(\mathrm{s})$
The Born-Haber cycle takes place as follows
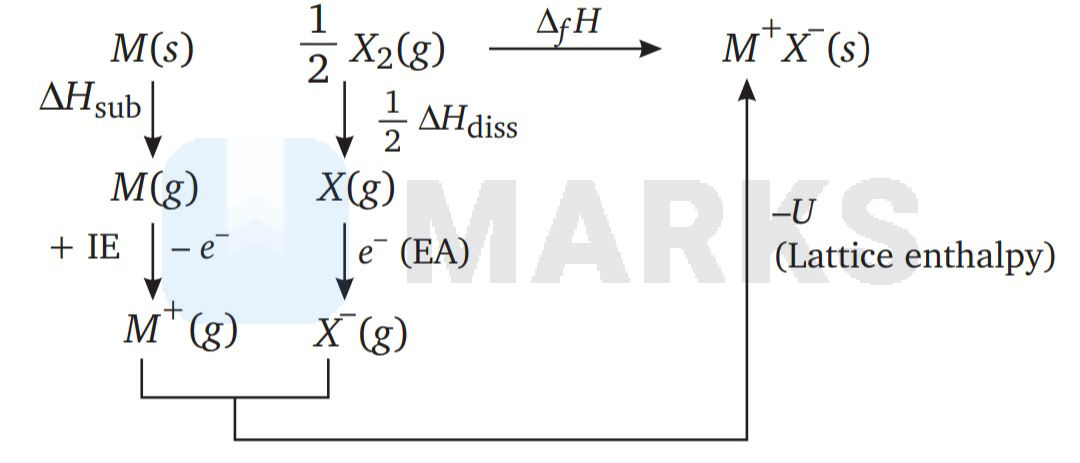
Hence, $\mathrm{Z}$ is $\mathrm{M}^{+} \mathrm{X}^{-}$(s).

Hence, $\mathrm{Z}$ is $\mathrm{M}^{+} \mathrm{X}^{-}$(s).
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.