Search any question & find its solution
Question:
Answered & Verified by Expert
Consider the change in oxidation state of Bromine corresponding to different emf values as
shown in the diagram below
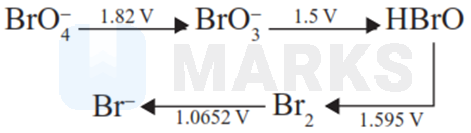
Then the species undergoing disproportionation is
Options:
shown in the diagram below
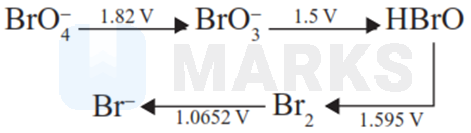
Then the species undergoing disproportionation is
Solution:
1004 Upvotes
Verified Answer
The correct answer is:
HBrO
for the disproportionation of HBrO,
Hence, option (C) is correct answer as is positive in it.
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.