Search any question & find its solution
Question:
Answered & Verified by Expert
Consider the following carbocations
The correct sequence of increasing order of their stabilities is
Options:
The correct sequence of increasing order of their stabilities is
Solution:
1564 Upvotes
Verified Answer
The correct answer is:
1 < 2 < 3 < 4
Resonance effect (+ M effect) are always more stabilizing than the inductive effects (+I effect), because +M groups neutralize the +ve charge on the carbon atom more effectively than the +I groups.
Triphenyl carbocation in the most stable in this series, because its +ve charge is dispersed by resonance (+M effect of group).
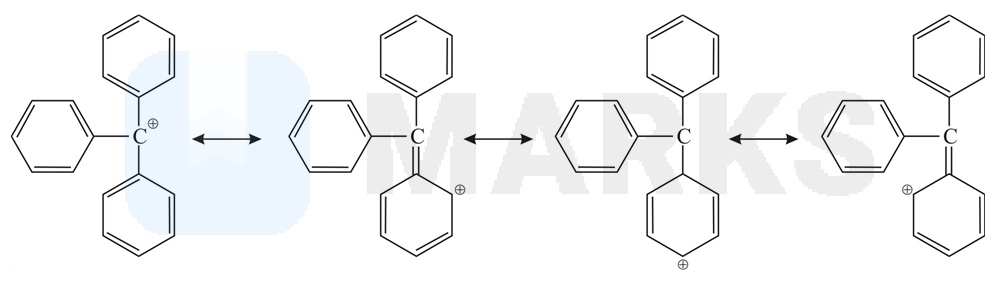
The correct order of stability is
1 < 2 < 3 < 4
Triphenyl carbocation in the most stable in this series, because its +ve charge is dispersed by resonance (+M effect of group).

The correct order of stability is
1 < 2 < 3 < 4
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.