Search any question & find its solution
Question:
Answered & Verified by Expert
Consider the following compounds:
$\text { (i) } \mathrm{C}_6 \mathrm{H}_5 \mathrm{COCl}$

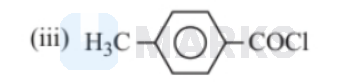
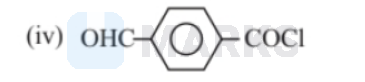
The correct decreasing order of their reactivity towards hydrolysis is:
Options:
$\text { (i) } \mathrm{C}_6 \mathrm{H}_5 \mathrm{COCl}$



The correct decreasing order of their reactivity towards hydrolysis is:
Solution:
2027 Upvotes
Verified Answer
The correct answer is:
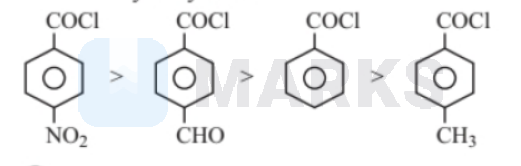
(ii) $>$ (iv) $>$ (i) $>$ (iii)
Electron withdrawing group increases the positive charge and electron releasing group decreases the negative change. The degree of hydrolysis increases as the magnitude of positive charge on carbonyl group increases. Among these $\mathrm{NO}_2$ \& $\mathrm{CHO}$ are electron withdrawing group from which $\mathrm{NO}_2$ has more $-\mathrm{I}$ effect than-CHO. On the other hand $\mathrm{CH}_3$ is a electron releasing group.
Thus, the order of reactivity towards hydrolysis is
Related Theory
The mesomeric effect is negative $(-M)$ when the substituent is an electron-withdrawing group and the effect is positive $(+M)$ when the substituent is an electron donating group.
Thus, the order of reactivity towards hydrolysis is

Related Theory
The mesomeric effect is negative $(-M)$ when the substituent is an electron-withdrawing group and the effect is positive $(+M)$ when the substituent is an electron donating group.
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.