Search any question & find its solution
Question:
Answered & Verified by Expert
Consider the following reaction sequence
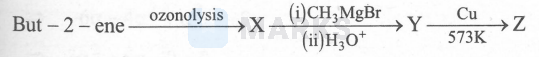
The correct statements about $\mathrm{Z}$ are
I. It gives yellow precipitate with $\mathrm{I}_2$ and $\mathrm{NaOH}$ solution
II. It undergoes disproportionation reaction in the presence of concentrated $\mathrm{NaOH}$ solution
III. It undergoes wolff-Kishner reduction
IV. It forms red precipitate with Fehling's reagent
Options:
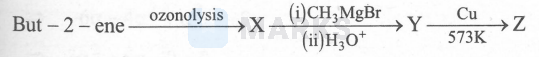
The correct statements about $\mathrm{Z}$ are
I. It gives yellow precipitate with $\mathrm{I}_2$ and $\mathrm{NaOH}$ solution
II. It undergoes disproportionation reaction in the presence of concentrated $\mathrm{NaOH}$ solution
III. It undergoes wolff-Kishner reduction
IV. It forms red precipitate with Fehling's reagent
Solution:
2823 Upvotes
Verified Answer
The correct answer is:
I & III only
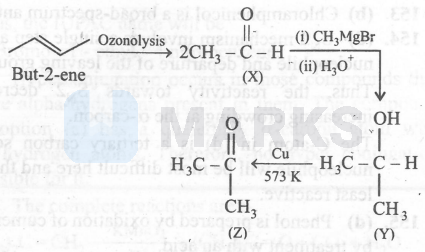
Since ' $\mathrm{Z}$ ' is a methyl ketone, it gives positive iodoform test and gives a yellow precipitate with $\mathrm{I}_2$ and $\mathrm{NaOH}$. Aldehydes and ketones undergo walff-kishner reduction to give the corresponding hydrocarbon.
Thus, I and III are correct.,
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.