Search any question & find its solution
Question:
Answered & Verified by Expert
How do you explain the amphoteric behaviour of amino acids?
Solution:
2627 Upvotes
Verified Answer
Amino acids contain an acidic (carboxyl group) and basic (amino group) group in the same molecule. In aqueous solution, they neutralize each other. The carboxyl group loses a proton while the amino group accepts it. As a result, a dipolar or zwitter ion is formed.
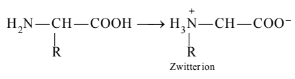
In zwitter ionic form, \(\alpha\)-amino acid show amphoteric behaviour as they react with both acids and bases.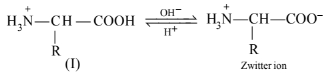
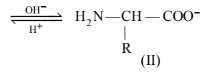
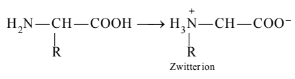
In zwitter ionic form, \(\alpha\)-amino acid show amphoteric behaviour as they react with both acids and bases.

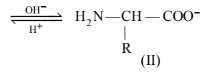
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.