Search any question & find its solution
Question:
Answered & Verified by Expert
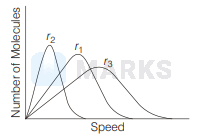
If $r_1, r_2$ and $r_3$ represent the most probable speeds of three different gases at the same temperature as shown in figure with molar masses $M_1, M_2$ and $M_3$ respectively. The correct order of molar masses of these gases is
Options:

Solution:
2803 Upvotes
Verified Answer
The correct answer is:
$M_2>M_1>M_3$
In the direction of $x$-axis $r_3$ increases and for same temperature most probable speed
$$
\propto \frac{1}{\sqrt{\text { molar mass }(M)}}
$$
Thus, $M_2>M_1>M_3$.
$$
\propto \frac{1}{\sqrt{\text { molar mass }(M)}}
$$
Thus, $M_2>M_1>M_3$.
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.