Search any question & find its solution
Question:
Answered & Verified by Expert
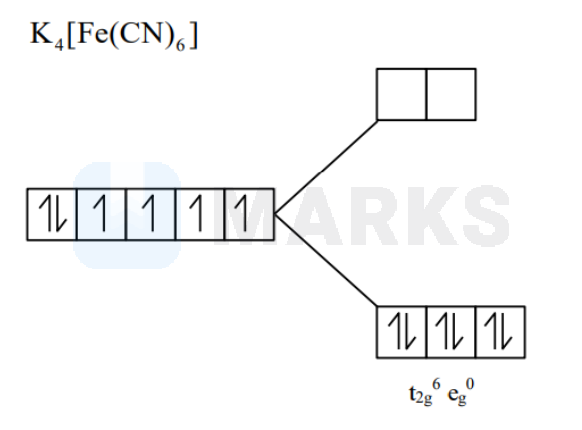
In potassium ferrocyanide, there are ______ pairs of electrons in the set of orbitals.
Solution:
1988 Upvotes
Verified Answer
The correct answer is:
3
Potassium ferrocyanide, , has a central iron ion that is coordinated with six cyanide ligands in an octahedral geometry. The oxidation state of the iron ion in potassium ferrocyanide is , hence has hybridised state. On applying . it shows that all the electrons of subshell are present in the form of three pairs in orbitals. Hence answer is .
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.