Search any question & find its solution
Question:
Answered & Verified by Expert
In the Claisen-Schmidt reaction to prepare \(351 \mathrm{~g}\) of dibenzalacetone using \(87 \mathrm{~g}\) of acetone, the amount of benzaldehyde required is ______ g. (Nearest integer)
Solution:
2255 Upvotes
Verified Answer
The correct answer is:
318
Claisen Schmidt reaction
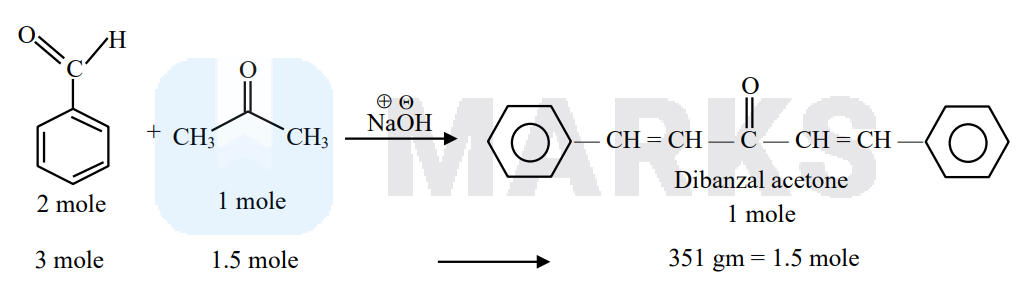
mw of benzaldehyde $=106$
$106 \times 3=318 \mathrm{gm}$. Benzaldehyde is required to give 1.5 mole (or $351 \mathrm{gm}$ ) product

mw of benzaldehyde $=106$
$106 \times 3=318 \mathrm{gm}$. Benzaldehyde is required to give 1.5 mole (or $351 \mathrm{gm}$ ) product
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.