Search any question & find its solution
Question:
Answered & Verified by Expert
In the following molecules,
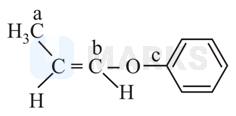
Hybridisation of carbon and respectively are :
Solution:
1583 Upvotes
Verified Answer
The correct answer is:
Hybridisation is found by steric number which is generally the summation of sigma bonds and lone pairs of electrons.
So as we can see has four sigma bonds so it has hybridisation.
has three sigma bonds so it has hybridisation.
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.