Search any question & find its solution
Question:
Answered & Verified by Expert
In the given reaction : $A+2 B \rightleftharpoons 2 C$ 2 moles each of $A$ and $B$ present in $10 \mathrm{~L}$ of solution combine to form 1 mole of $C$. Calculate $K_c$ for the reaction.
Options:
Solution:
2580 Upvotes
Verified Answer
The correct answer is:
$6.67$
$A+2 B \rightleftharpoons 2 C$
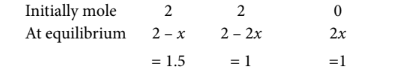
As 1 mole of $C$ is formed at equilibrium,
$2 x=1 \Rightarrow x=0.5$
$\Rightarrow$ Number of moles of $A$ at equilibrium $=1.5, B=1$ and $C=1$
$\therefore$ Equilibrium concentration of $A=1.5 / 10$
$\begin{gathered}B=1 / 10 \text { and } C=1 / 10 \\ K_c=\frac{[C]^2}{[A][B]^2}=\frac{[1 / 10]^2}{[1.5 / 10][1 / 10]^2}=6.67\end{gathered}$
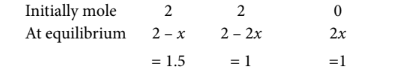
As 1 mole of $C$ is formed at equilibrium,
$2 x=1 \Rightarrow x=0.5$
$\Rightarrow$ Number of moles of $A$ at equilibrium $=1.5, B=1$ and $C=1$
$\therefore$ Equilibrium concentration of $A=1.5 / 10$
$\begin{gathered}B=1 / 10 \text { and } C=1 / 10 \\ K_c=\frac{[C]^2}{[A][B]^2}=\frac{[1 / 10]^2}{[1.5 / 10][1 / 10]^2}=6.67\end{gathered}$
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.