Search any question & find its solution
Question:
Answered & Verified by Expert
In which of the following carbohydrate, molecular mass increases by 84 u after complete acetylation?
Options:
Solution:
1272 Upvotes
Verified Answer
The correct answer is:
Aldotriose
In acetylation reaction $\mathrm{H}$ atom of an $(-\mathrm{OH})$ group is replaced by an acetyl group $\left(-\mathrm{COCH}_3\right)$. This results increase in molecular mass by $[(12+16+12+3 \times 1)-1]$, that as, $42 \mathrm{u}$.
Increase in molecular mass $=84 \mathrm{u}$
$\therefore \quad$ Number of $-\mathrm{OH}$ groups $=\frac{84 \mathrm{u}}{42 \mathrm{u}}=2$
The carbohydrate must be aldotriose as it contains two alcoholic groups.
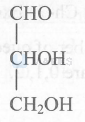
Increase in molecular mass $=84 \mathrm{u}$
$\therefore \quad$ Number of $-\mathrm{OH}$ groups $=\frac{84 \mathrm{u}}{42 \mathrm{u}}=2$
The carbohydrate must be aldotriose as it contains two alcoholic groups.
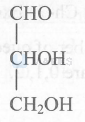
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.