Search any question & find its solution
Question:
Answered & Verified by Expert
Increasing order of basic strength among given compounds is-
Options:




Solution:
1020 Upvotes
Verified Answer
The correct answer is:
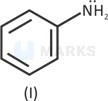
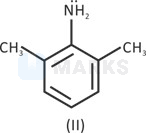
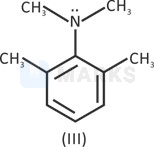
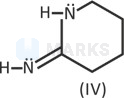
IV > III > I > II
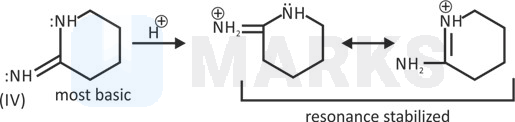
Its conjugate acid do not undergo proper solvation and hence its stability decreases. Hence,basic strength decreases.
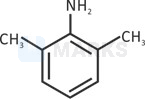
 Lone pair is in resonance.
Lone pair is in resonance.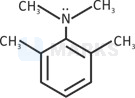
Steric inhibition(two methyl groups present at ortho position pushes nitrogen out of the plane of the benzene ring and prevents resonance of lone pair) takes place in resonance. (Basic strength increases).
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.