Search any question & find its solution
Question:
Answered & Verified by Expert
Increasing order of carbon-carbon bond length for the following is
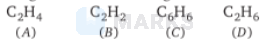
Options:
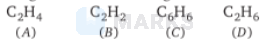
Solution:
1505 Upvotes
Verified Answer
The correct answer is:
$B < A < C < D$
In $\mathrm{C}_{2} \mathrm{H}_{6}$, carbon-carbon bond is single (1.54 А). In $\mathrm{C}_{6} \mathrm{H}_{6}$, carbon-carbon bond is partially double (1.39 A)
In $\mathrm{C}_{2} \mathrm{H}_{4}$, carbon-carbon bond is double $(1.34 Å)$
In $\mathrm{C}_{2} \mathrm{H}_{2}$, carbon-carbon bond is triple $(1.10 Å)$
In $\mathrm{C}_{2} \mathrm{H}_{4}$, carbon-carbon bond is double $(1.34 Å)$
In $\mathrm{C}_{2} \mathrm{H}_{2}$, carbon-carbon bond is triple $(1.10 Å)$
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.