Search any question & find its solution
Question:
Answered & Verified by Expert
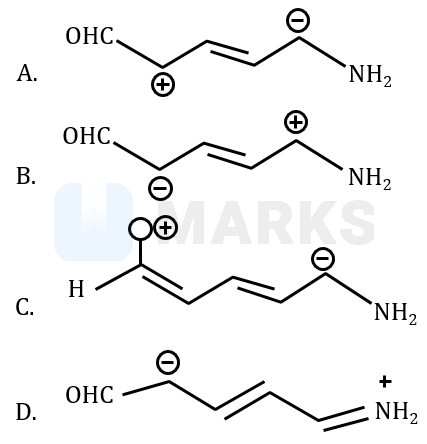
Increasing order of stability of the resonance structure is:
Solution:
2202 Upvotes
Verified Answer
The correct answer is:
In the molecule C, positive charge is present on the electronegative oxygen atom and is not having complete octet. Hence, the structure C is the least stable resonance structure. In the structure A, the delocalisation is taken place away from the electron withdrawing group, which is not favourable delocalisation. In the structure D, more number of covalent bonds are there and having all atoms' octet.
Order should be :
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.