Search any question & find its solution
Question:
Answered & Verified by Expert
Indicate the steps in the preparation of:
(i) \(\mathrm{K}_2 \mathrm{Cr}_2 \mathrm{O}_7\) from chromite ore
(ii) \(\mathrm{KMnO}_4\) from pyrolusite ore.
(i) \(\mathrm{K}_2 \mathrm{Cr}_2 \mathrm{O}_7\) from chromite ore
(ii) \(\mathrm{KMnO}_4\) from pyrolusite ore.
Solution:
2233 Upvotes
Verified Answer
(i) \(4 \mathrm{FeCr}_2 \mathrm{O}_4+8 \mathrm{Na}_2 \mathrm{CO}_3+7 \mathrm{O}_2 \rightarrow\)
\(\begin{aligned}
8 \mathrm{Na}_2 \mathrm{CrO}_4+2 \mathrm{Fe}_2 \mathrm{O}_3+8 \mathrm{CO}_2 \\
2 \mathrm{Na}_2 \mathrm{CrO}_4+2 \mathrm{H}^{+} \rightarrow \mathrm{Na}_2 \mathrm{Cr}_2 \mathrm{O}_7+2 \mathrm{Na}^{+}+\mathrm{H}_2 \mathrm{O} \\
\mathrm{Na}_2 \mathrm{Cr}_2 \mathrm{O}_7+2 \mathrm{KCl} \rightarrow \mathrm{K}_2 \mathrm{Cr}_2 \mathrm{O}_7+2 \mathrm{NaCl}
\end{aligned}\)
(ii) \(2 \mathrm{MnO}_2+4 \mathrm{KOH}+\mathrm{O}_2 \stackrel{\Delta}{\longrightarrow} 2 \mathrm{~K}_2 \mathrm{MnO}_4+2 \mathrm{H}_2 \mathrm{O}\)
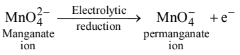
\(\begin{aligned}
8 \mathrm{Na}_2 \mathrm{CrO}_4+2 \mathrm{Fe}_2 \mathrm{O}_3+8 \mathrm{CO}_2 \\
2 \mathrm{Na}_2 \mathrm{CrO}_4+2 \mathrm{H}^{+} \rightarrow \mathrm{Na}_2 \mathrm{Cr}_2 \mathrm{O}_7+2 \mathrm{Na}^{+}+\mathrm{H}_2 \mathrm{O} \\
\mathrm{Na}_2 \mathrm{Cr}_2 \mathrm{O}_7+2 \mathrm{KCl} \rightarrow \mathrm{K}_2 \mathrm{Cr}_2 \mathrm{O}_7+2 \mathrm{NaCl}
\end{aligned}\)
(ii) \(2 \mathrm{MnO}_2+4 \mathrm{KOH}+\mathrm{O}_2 \stackrel{\Delta}{\longrightarrow} 2 \mathrm{~K}_2 \mathrm{MnO}_4+2 \mathrm{H}_2 \mathrm{O}\)

Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.