Search any question & find its solution
Question:
Answered & Verified by Expert
\(\left[\mathrm{Co}\left(\mathrm{NH}_3\right)_4\left(\mathrm{NO}_2\right)_2\right] \mathrm{Cl}\) exhibits
Options:
Solution:
1241 Upvotes
Verified Answer
The correct answer is:
linkage isomerism, ionization isomerism and geometrical isomerism
The given compound may have linkage isomerism due to presence of \(\mathrm{NO}_2\) group which may be in the form \(-\mathrm{NO}_2\) or \(-\mathrm{ONO}\).
It may have ionisation isomerism due to presence of two ionisable group \(-\mathrm{NO}_2 \&-\mathrm{Cl}\). It may have geometrical isomerism in the form of cis-trans form as follows :
\(\left[\mathrm{Co}\left(\mathrm{NH}_3\right)_4 \mathrm{Cl}\left(\mathrm{NO}_2\right)\right] \mathrm{NO}_2 \&\left[\mathrm{Co}\left(\mathrm{NH}_3\right)\left(\mathrm{NO}_2\right)_2\right] \mathrm{Cl}-\) - ionisation isomers.
\(\left[\mathrm{Co}\left(\mathrm{NH}_3\right)_5\left(\mathrm{NO}_2\right)_2\right] \mathrm{Cl} \&\left[\mathrm{Co}\left(\mathrm{NH}_3\right)_5(\mathrm{ONO})_2 \mathrm{Cl}\right.\) -Linkage isomers
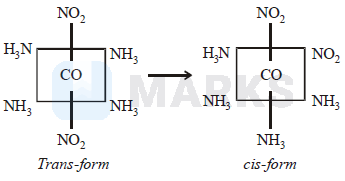
Geometrical isomers
It may have ionisation isomerism due to presence of two ionisable group \(-\mathrm{NO}_2 \&-\mathrm{Cl}\). It may have geometrical isomerism in the form of cis-trans form as follows :
\(\left[\mathrm{Co}\left(\mathrm{NH}_3\right)_4 \mathrm{Cl}\left(\mathrm{NO}_2\right)\right] \mathrm{NO}_2 \&\left[\mathrm{Co}\left(\mathrm{NH}_3\right)\left(\mathrm{NO}_2\right)_2\right] \mathrm{Cl}-\) - ionisation isomers.
\(\left[\mathrm{Co}\left(\mathrm{NH}_3\right)_5\left(\mathrm{NO}_2\right)_2\right] \mathrm{Cl} \&\left[\mathrm{Co}\left(\mathrm{NH}_3\right)_5(\mathrm{ONO})_2 \mathrm{Cl}\right.\) -Linkage isomers

Geometrical isomers
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.