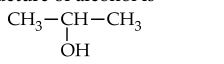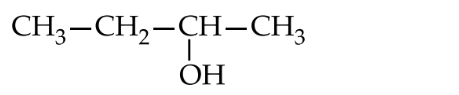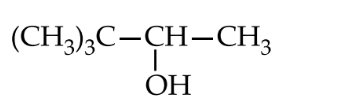Search any question & find its solution
Question:
Answered & Verified by Expert
Lucas test is given by an alcohol within 5 minutes. $0.22 \mathrm{~g}$ of which liberates $56 \mathrm{~mL}$ of $\mathrm{CH}_4$ at STP on treating with $\mathrm{CH}_3$ MgI. The structure of alcohol is
Options:
Solution:
2019 Upvotes
Verified Answer
The correct answer is:
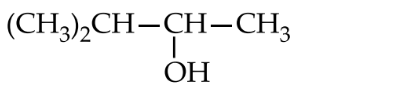
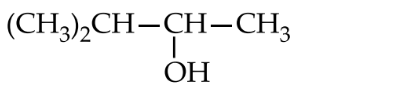
Lucas test will be given by all the given alcohols within 5 minutes as all are $2^{\circ}$ alcohols. Exact structure can be known by molecular mass or formula of alcohol which will be obtained as follows :
Let molecular mass of alcohol be $M$.
$\frac{56}{22400}=\frac{0.22}{M}$ or, $M=\frac{22400 \times 0.22}{56}=88$
The general formula of alcohols is $\mathrm{C}_n \mathrm{H}_{2 n+1} \mathrm{OH}$. The molecular mass 88 corresponds to the value of $n=5$. Thus, the $2^{\circ}$ alcohol is $\left(\mathrm{CH}_3\right)_2 \mathrm{CHCHOHCH}_3$.
Let molecular mass of alcohol be $M$.
$\frac{56}{22400}=\frac{0.22}{M}$ or, $M=\frac{22400 \times 0.22}{56}=88$
The general formula of alcohols is $\mathrm{C}_n \mathrm{H}_{2 n+1} \mathrm{OH}$. The molecular mass 88 corresponds to the value of $n=5$. Thus, the $2^{\circ}$ alcohol is $\left(\mathrm{CH}_3\right)_2 \mathrm{CHCHOHCH}_3$.
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.