Search any question & find its solution
Question:
Answered & Verified by Expert
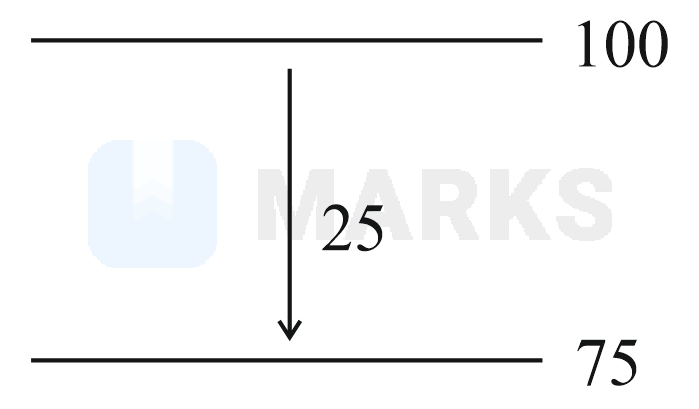
Mass of Urea required to be dissolved in of water in order to reduce the vapour pressure of water by is (Nearest integer)
Given : Molar mass of and are and respectively.
Solution:
1413 Upvotes
Verified Answer
The correct answer is:
1111
Relative lowering of vapour pressureMole fraction of solute
moles of urea
Mass of urea
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.