Search any question & find its solution
Question:
Answered & Verified by Expert
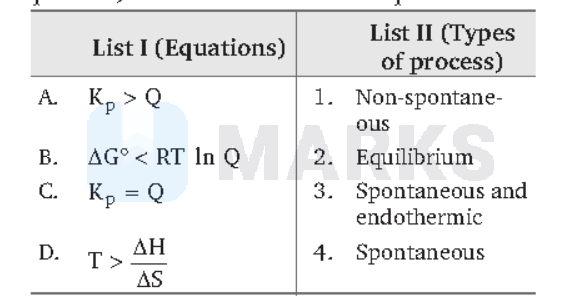
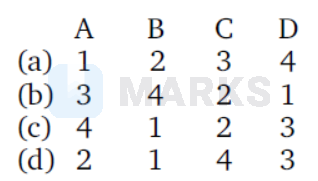
Match List I (equations) with List II (types of process) and select the correct OPTION.
Options:


Solution:
2035 Upvotes
Verified Answer
The correct answer is:
(c)
(A) When $K_p > Q$, the reaction goes in forward direction, ie, the reaction is spontaneous.
(B) Given, $\Delta G^{\circ} < R T \ln Q$, thus, $\Delta \mathrm{G}^{\circ}=+$ ve and hence, the reaction is nonspontaneous.
(C) At equilibrium, $\mathrm{K}_{\mathrm{p}}=\mathrm{Q}$
(D) $\mathrm{T} > \frac{\Delta \mathrm{H}}{\Delta \mathrm{S}}$ or $\mathrm{T} \Delta \mathrm{S} > \Delta \mathrm{H}$
This condition is true for spontaneous endothermic reactions (as $\Delta G \geq \Delta \mathrm{H}-\mathrm{T} \Delta \mathrm{S}$).
(B) Given, $\Delta G^{\circ} < R T \ln Q$, thus, $\Delta \mathrm{G}^{\circ}=+$ ve and hence, the reaction is nonspontaneous.
(C) At equilibrium, $\mathrm{K}_{\mathrm{p}}=\mathrm{Q}$
(D) $\mathrm{T} > \frac{\Delta \mathrm{H}}{\Delta \mathrm{S}}$ or $\mathrm{T} \Delta \mathrm{S} > \Delta \mathrm{H}$
This condition is true for spontaneous endothermic reactions (as $\Delta G \geq \Delta \mathrm{H}-\mathrm{T} \Delta \mathrm{S}$).
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.