Search any question & find its solution
Choose the correct answer from the options given below :-
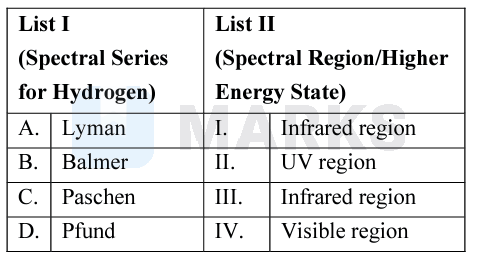
The Lyman series lies in the ultraviolet, in chemistry, the Lyman series is a hydrogen spectral series of transitions and resulting ultraviolet emission lines of the hydrogen atom as an electron goes from to (where is the principal quantum number), the lowest energy level of the electron.
All of the wavelengths in the Balmer series are in the visible region of the electromagnetic spectrum ( to ). The presence of Hydrogen is detected in astronomy by using the Alpha line of the Balmer series; it is also a component of the solar spectrum.
All the wavelength of the Paschen series falls in the Infrared region of the electromagnetic spectrum.
Pfund series lie in the infrared.
A – II, B – IV, C – III, D – I
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.