Search any question & find its solution
Question:
Answered & Verified by Expert
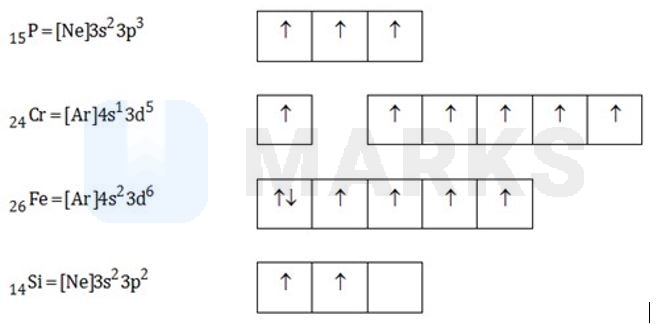
Match the column I with column II and mark the appropriate choice.
Options:
| Column I | Column II | ||
| (p) | (i) | 6 unpaired electrons | |
| (q) | (ii) | 2 unpaired electrons | |
| (r) | (iii) | 3 unpaired electrons | |
| (s) | (iv) | 4 unpaired electrons |
Solution:
2114 Upvotes
Verified Answer
The correct answer is:
(p)-(iii), (q)-(i), (r)-(iv), (s)-(ii)
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.