Search any question & find its solution
Question:
Answered & Verified by Expert
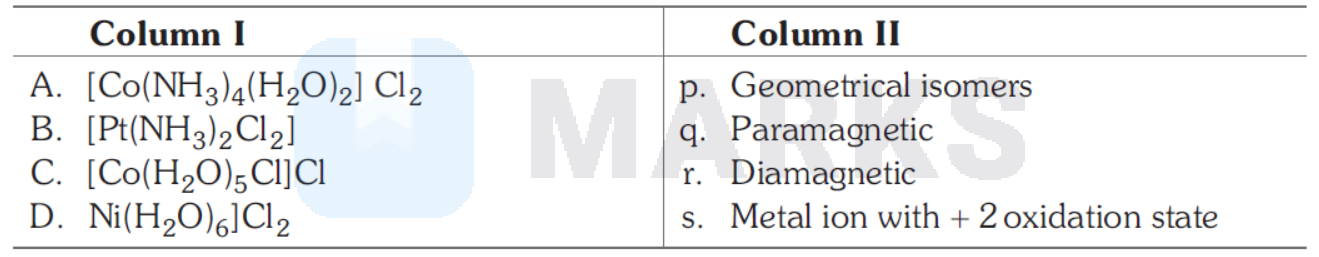
Match the complexes in Column I with their properties listed in Column II. Indicates your answer by darkening the appropriate bubbles of $4 \times 4$ matrix given in the ORS.
Options:

Solution:
2122 Upvotes
Verified Answer
The correct answer is:
A-p, q, s; B-p, r, s; C-q, s; D-q, s
A-p, q, s; B-p, r, s; C-q, s; D-q, s
(A) $\left[\mathrm{Co}\left(\mathrm{NH}_3\right)_4\left[\left(\mathrm{H}_2 \mathrm{O}\right)_2\right] \mathrm{Cl}_2 p, q, s\right.$
Oxidation state $\mathrm{Co}$ is $+2$
$\Rightarrow$ Paramagnetic, exhibits geometrical isomerism.
(B) $\left[\mathrm{Pt}\left(\mathrm{NH}_3\right)_4 \mathrm{Cl}_2\right] p, r, s$
$\mathrm{dsp}{ }^2$-hybridisation
$\mathrm{Pt}^{2+}$, diamagnetic square planar, geometrical isomerism
(C) $q, s$ (D) $q, s$
Oxidation state $\mathrm{Co}$ is $+2$
$\Rightarrow$ Paramagnetic, exhibits geometrical isomerism.
(B) $\left[\mathrm{Pt}\left(\mathrm{NH}_3\right)_4 \mathrm{Cl}_2\right] p, r, s$
$\mathrm{dsp}{ }^2$-hybridisation
$\mathrm{Pt}^{2+}$, diamagnetic square planar, geometrical isomerism
(C) $q, s$ (D) $q, s$
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.