Search any question & find its solution
Question:
Answered & Verified by Expert
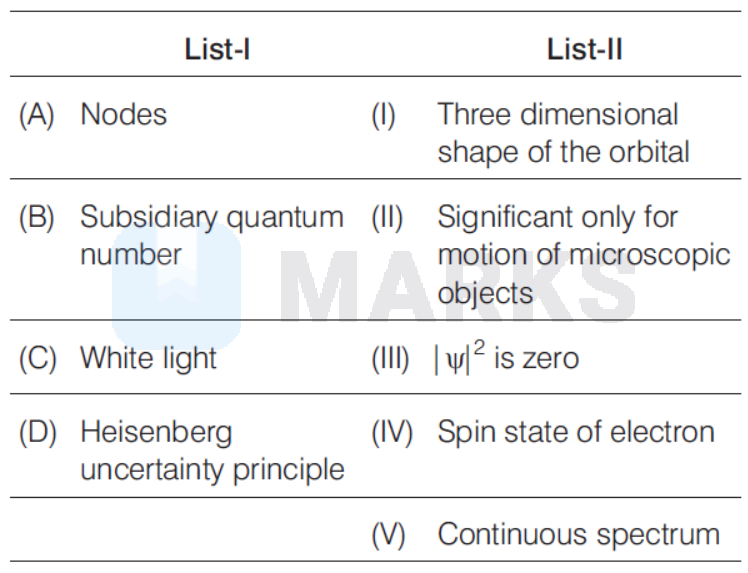
Match the following.

Options:


Solution:
2351 Upvotes
Verified Answer
The correct answer is:
(d)
(A) Nodes (III) $\left|\psi^2\right|$ is zero because $\left|\psi^2\right|$ represents the region where probability of electron finding is zero.
(B) Subsidiary Quantum Number
(I) is a three dimensional shape of the orbital. It is also called azimuthal quantum number (1). The values of (l) gives three dimensional shapes of orbitals.
(C) White Light
(V) Continuous spectrum because when all the energy waves are so close to each other that they appear as in continuous pattern, the spectrum is called continuous spectrum.
(D) (II) Heisenberg's Uncertainty Principle
Significant only for motion of microscopic objects, because, according to this principle, it is impossible to determine simultaneously, the exact position and exact momentum (or velocity) of an electron. Hence, (d) is the correct option.
(B) Subsidiary Quantum Number
(I) is a three dimensional shape of the orbital. It is also called azimuthal quantum number (1). The values of (l) gives three dimensional shapes of orbitals.
(C) White Light
(V) Continuous spectrum because when all the energy waves are so close to each other that they appear as in continuous pattern, the spectrum is called continuous spectrum.
(D) (II) Heisenberg's Uncertainty Principle
Significant only for motion of microscopic objects, because, according to this principle, it is impossible to determine simultaneously, the exact position and exact momentum (or velocity) of an electron. Hence, (d) is the correct option.
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.