Search any question & find its solution
Question:
Answered & Verified by Expert
Match the following:
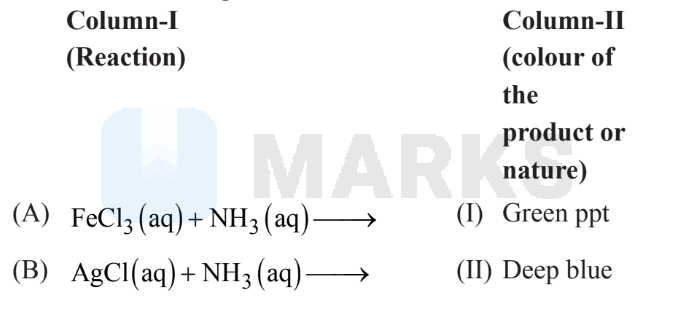
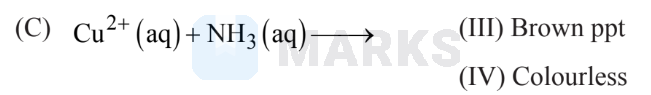
The correct answer is
$$
\begin{array}{lll}
\text { A } & \text { B } & \text { C }
\end{array}
$$
Options:
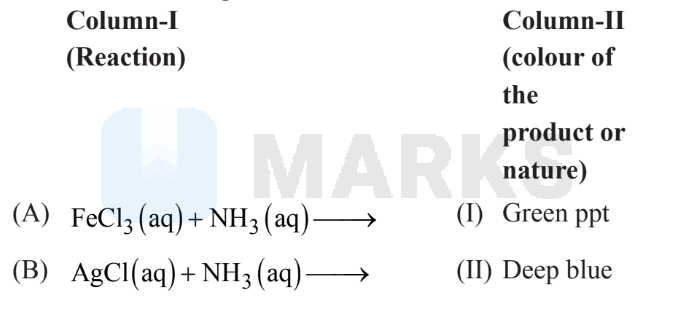
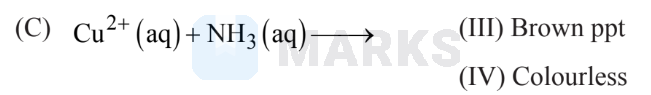
The correct answer is
$$
\begin{array}{lll}
\text { A } & \text { B } & \text { C }
\end{array}
$$
Solution:
1124 Upvotes
Verified Answer
The correct answer is:
III IV II
$\mathrm{Cu}^{2+}+4 \mathrm{NH}_3(\mathrm{aq}) \rightarrow\left[\mathrm{Cu}\left(\mathrm{NH}_3\right)_4\right]^{2+}$
(Deep blue aqueous soln.)
$$
\begin{aligned}
& \mathrm{AgCl}+\mathrm{NH}_3(\mathrm{aq}) \rightarrow\left[\mathrm{Ag}\left(\mathrm{NH}_3\right)_2\right] \mathrm{Cl} \quad \text { (Colourless) } \\
& \mathrm{FeCl}_3+\mathrm{NH}_4 \mathrm{OH} \rightarrow \mathrm{Fe}_2 \mathrm{O}_3 \cdot \mathrm{xH}_2 \mathrm{O}+\mathrm{NH}_4 \mathrm{Cl}(\mathrm{aq})
\end{aligned}
$$
(Brown ppt.)
(Deep blue aqueous soln.)
$$
\begin{aligned}
& \mathrm{AgCl}+\mathrm{NH}_3(\mathrm{aq}) \rightarrow\left[\mathrm{Ag}\left(\mathrm{NH}_3\right)_2\right] \mathrm{Cl} \quad \text { (Colourless) } \\
& \mathrm{FeCl}_3+\mathrm{NH}_4 \mathrm{OH} \rightarrow \mathrm{Fe}_2 \mathrm{O}_3 \cdot \mathrm{xH}_2 \mathrm{O}+\mathrm{NH}_4 \mathrm{Cl}(\mathrm{aq})
\end{aligned}
$$
(Brown ppt.)
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.