Search any question & find its solution
Question:
Answered & Verified by Expert
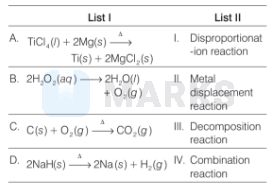
Match the following:
The correct answer is
$\begin{array}{llllllll}A & B & C & D & A & B & C & D\end{array}$
Options:

The correct answer is
$\begin{array}{llllllll}A & B & C & D & A & B & C & D\end{array}$
Solution:
1146 Upvotes
Verified Answer
The correct answer is:
$\begin{array}{llll}\| & \text { I IV } & \text { III }\end{array}$
(A) $\mathrm{TiCl}_4(l)+2 \mathrm{Mg}(s) \stackrel{\Delta}{\longrightarrow} \mathrm{Ti}(s)+2 \mathrm{MgCl}_2(s)$
The above reaction is an example of metal displacement reaction in which more reactive metal. (i.e. $\mathrm{Mg}$ ) will displace less reactive metal (i.e. Ti).
Thus, option (II) is related with (A).
(B) $2 \mathrm{H}_2 \mathrm{O}_2(a q) \longrightarrow 2 \mathrm{H}_2 \mathrm{O}(l)+\mathrm{O}_2(g)$
The above reaction is an example of disproportionation reaction in which $\mathrm{O}$-atom of $\mathrm{H}_2 \mathrm{O}_2$ is oxidised as $\left(\mathrm{O}_2\right)$ and also reduced to $\mathrm{H}_2 \mathrm{O}$. Thus, option (I) is related with (B).
(C) $\mathrm{C}(s)+\mathrm{O}_2(g) \stackrel{\Delta}{\longrightarrow} \mathrm{CO}_2(g)$
The above reaction is an example of combination reaction in which carbon(s) combines with $\mathrm{O}_2(g)$ to give $\mathrm{CO}_2(g)$. Thus, option (IV) is related with (C).
(D) $2 \mathrm{NaH}(s) \stackrel{\Delta}{\longrightarrow} 2 \mathrm{Na}(s)+\mathrm{H}_2(g)$
The above reaction is an example of decomposition reaction in which $\mathrm{NaH}$ is decomposes to $\mathrm{Na}(s)$ and $\mathrm{H}_2(g)$. Thus, option (III) is related with (D).
Hence,
$(\mathrm{A}) \rightarrow(\mathrm{II}),(\mathrm{B}) \rightarrow(\mathrm{I}),(\mathrm{C}) \rightarrow(\mathrm{IV}),(\mathrm{D}) \rightarrow(\mathrm{III})$
and option (a) is the correct answer.
The above reaction is an example of metal displacement reaction in which more reactive metal. (i.e. $\mathrm{Mg}$ ) will displace less reactive metal (i.e. Ti).
Thus, option (II) is related with (A).
(B) $2 \mathrm{H}_2 \mathrm{O}_2(a q) \longrightarrow 2 \mathrm{H}_2 \mathrm{O}(l)+\mathrm{O}_2(g)$
The above reaction is an example of disproportionation reaction in which $\mathrm{O}$-atom of $\mathrm{H}_2 \mathrm{O}_2$ is oxidised as $\left(\mathrm{O}_2\right)$ and also reduced to $\mathrm{H}_2 \mathrm{O}$. Thus, option (I) is related with (B).
(C) $\mathrm{C}(s)+\mathrm{O}_2(g) \stackrel{\Delta}{\longrightarrow} \mathrm{CO}_2(g)$
The above reaction is an example of combination reaction in which carbon(s) combines with $\mathrm{O}_2(g)$ to give $\mathrm{CO}_2(g)$. Thus, option (IV) is related with (C).
(D) $2 \mathrm{NaH}(s) \stackrel{\Delta}{\longrightarrow} 2 \mathrm{Na}(s)+\mathrm{H}_2(g)$
The above reaction is an example of decomposition reaction in which $\mathrm{NaH}$ is decomposes to $\mathrm{Na}(s)$ and $\mathrm{H}_2(g)$. Thus, option (III) is related with (D).
Hence,
$(\mathrm{A}) \rightarrow(\mathrm{II}),(\mathrm{B}) \rightarrow(\mathrm{I}),(\mathrm{C}) \rightarrow(\mathrm{IV}),(\mathrm{D}) \rightarrow(\mathrm{III})$
and option (a) is the correct answer.
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.