Search any question & find its solution
Question:
Answered & Verified by Expert
Match the following.
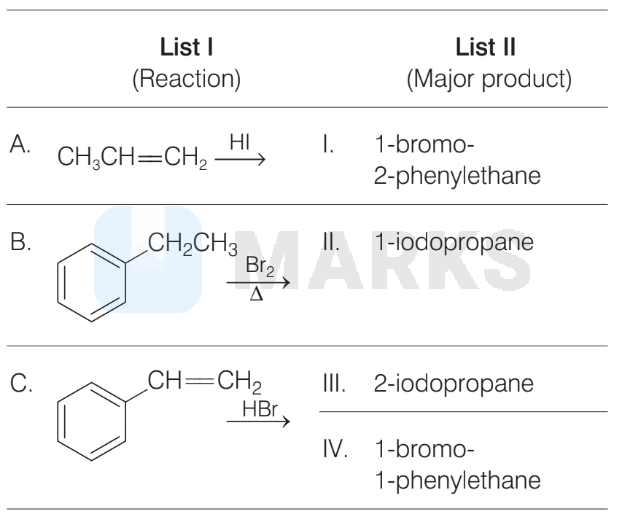
The correct match is
$\text { A } \quad B \quad C$
Options:

The correct match is
$\text { A } \quad B \quad C$
Solution:
2563 Upvotes
Verified Answer
The correct answer is:
III IV IV
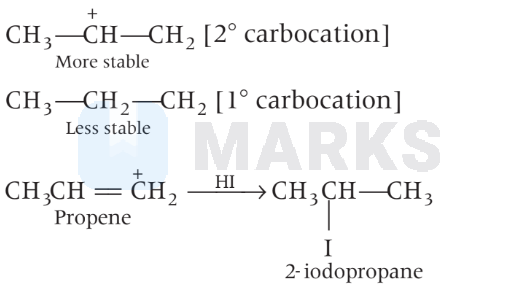
(A) Addition of HI takes place by the formation of $1^{\circ}$ and $2^{\circ}$ carbonium ion. $2^{\circ}$ carbonium ion being more stable will form major product, i.e. 2-iodopropane.
(B) The reaction of bromine with 1-phenylethane proceed with formation of free radical. $2^{\circ}$ free radical being more stable than $1^{\circ}$ free radical, will form the product as 1-bromo-1-phenylethane.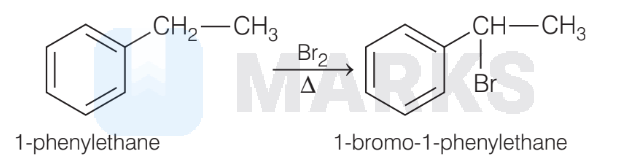
(C) Reaction of $\mathrm{HBr}$ to an alkene follows Markownikoff rule. So, the product will be 1-bromo-1-phenylethane.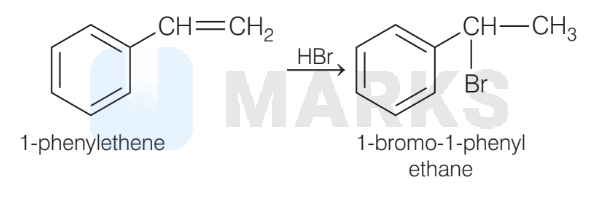
Note : Markownikoff's rule states that, electron rich component adds to carbon atom with less number of hydrogen atoms.
Hence, the correct match is A-III, B-IV, C-IV. The reactions are :

(B) The reaction of bromine with 1-phenylethane proceed with formation of free radical. $2^{\circ}$ free radical being more stable than $1^{\circ}$ free radical, will form the product as 1-bromo-1-phenylethane.

(C) Reaction of $\mathrm{HBr}$ to an alkene follows Markownikoff rule. So, the product will be 1-bromo-1-phenylethane.

Note : Markownikoff's rule states that, electron rich component adds to carbon atom with less number of hydrogen atoms.
Hence, the correct match is A-III, B-IV, C-IV. The reactions are :
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.