Search any question & find its solution
Question:
Answered & Verified by Expert
Match the following :
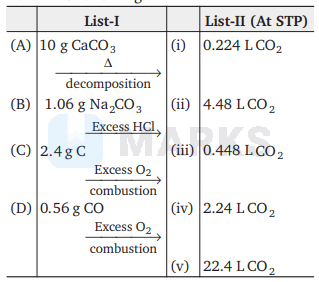
The correct match is
$\begin{array}{llll}\mathrm{A} & \mathrm{B} & \mathrm{C} & \mathrm{D}\end{array}$
Options:

The correct match is
$\begin{array}{llll}\mathrm{A} & \mathrm{B} & \mathrm{C} & \mathrm{D}\end{array}$
Solution:
2892 Upvotes
Verified Answer
The correct answer is:
$\begin{array}{llll}\text { iv } & \text { i } & \text { ii } & \text { iii }\end{array}$
(A)
$$
\begin{aligned}
& \underset{100 \mathrm{~g}}{\mathrm{CaCO}_3} \underset{\text { decomposition }}{\longrightarrow} \mathrm{CaO}+\underset{22.4 \mathrm{~L}}{\mathrm{CO}_2} \\
& \because 100 \mathrm{~g} \mathrm{CaCO}_3 \text { on decomposition gives } \\
& =22.4 \mathrm{~L} \mathrm{CO}_2 \\
& =\frac{22.4 \times 10}{100} \mathrm{LCO}_2 \\
& =2.24 \mathrm{~L} \mathrm{CO}_2 \\
&
\end{aligned}
$$
$\therefore 10 \mathrm{~g} \mathrm{CaCO}_3$ on decomposition will give
$$
\begin{aligned}
& =\frac{22.4 \times 10}{100} \mathrm{LCO}_2 \\
& =2.24 \mathrm{LCO}_2
\end{aligned}
$$
(B)
$$
\begin{aligned}
& \mathrm{Na}_2 \mathrm{CO}_3 \stackrel{\text { Excess } \mathrm{HCl}}{\longrightarrow} 2 \mathrm{NaCl}+\mathrm{H}_2 \mathrm{O}+\mathrm{CO}_2 \\
& 106 \mathrm{~g} \\
& 22.4 \mathrm{~L} \\
& 106 \mathrm{~g} \mathrm{Na}_2 \mathrm{CO}_3 \text { gives }=22.4 \mathrm{~L} \mathrm{CO}_2 \\
& 1.06 \mathrm{~g} \mathrm{Na}_2 \mathrm{CO}_3 \text { will give } \\
& =\frac{22.4 \times 1.06}{106} \mathrm{~L} \mathrm{CO}_2 \\
& =0.224 \mathrm{~L} \mathrm{CO}_2 \\
&
\end{aligned}
$$
$$
\begin{aligned}
& =\frac{22.4 \times 1.06}{106} \mathrm{LCO}_2 \\
& =0.224 \mathrm{~L} \mathrm{CO}_2
\end{aligned}
$$
(C)
$12 \mathrm{~g}$ carbon on combustion gives
$$
=22.4 \mathrm{~L} \mathrm{CO}_2
$$
$2.4 \mathrm{~g}$ carbon on combustion will give
$$
\begin{aligned}
& =\frac{22.4 \times 2.4}{12} \mathrm{LCO}_2 \\
& =2 \times 2.24 \mathrm{LCO}_2 \\
& =4.48 \mathrm{LCO}_2
\end{aligned}
$$
(D)
$56 \mathrm{~g}$ carbon monoxide on combustion gives $=2 \times 22.4 \mathrm{~L} \mathrm{CO}_2$
$0.56 \mathrm{~g}$ carbon monoxide on combustion will give
$$
\begin{aligned}
& =\frac{2 \times 22.4 \times 0.56}{56} \mathrm{LCO}_2 \\
& =0.448 \mathrm{LCO}_2
\end{aligned}
$$
Hence, A-(iv), B-(i), C-(ii), D-(iii)
$$
\begin{aligned}
& \underset{100 \mathrm{~g}}{\mathrm{CaCO}_3} \underset{\text { decomposition }}{\longrightarrow} \mathrm{CaO}+\underset{22.4 \mathrm{~L}}{\mathrm{CO}_2} \\
& \because 100 \mathrm{~g} \mathrm{CaCO}_3 \text { on decomposition gives } \\
& =22.4 \mathrm{~L} \mathrm{CO}_2 \\
& =\frac{22.4 \times 10}{100} \mathrm{LCO}_2 \\
& =2.24 \mathrm{~L} \mathrm{CO}_2 \\
&
\end{aligned}
$$
$\therefore 10 \mathrm{~g} \mathrm{CaCO}_3$ on decomposition will give
$$
\begin{aligned}
& =\frac{22.4 \times 10}{100} \mathrm{LCO}_2 \\
& =2.24 \mathrm{LCO}_2
\end{aligned}
$$
(B)
$$
\begin{aligned}
& \mathrm{Na}_2 \mathrm{CO}_3 \stackrel{\text { Excess } \mathrm{HCl}}{\longrightarrow} 2 \mathrm{NaCl}+\mathrm{H}_2 \mathrm{O}+\mathrm{CO}_2 \\
& 106 \mathrm{~g} \\
& 22.4 \mathrm{~L} \\
& 106 \mathrm{~g} \mathrm{Na}_2 \mathrm{CO}_3 \text { gives }=22.4 \mathrm{~L} \mathrm{CO}_2 \\
& 1.06 \mathrm{~g} \mathrm{Na}_2 \mathrm{CO}_3 \text { will give } \\
& =\frac{22.4 \times 1.06}{106} \mathrm{~L} \mathrm{CO}_2 \\
& =0.224 \mathrm{~L} \mathrm{CO}_2 \\
&
\end{aligned}
$$
$$
\begin{aligned}
& =\frac{22.4 \times 1.06}{106} \mathrm{LCO}_2 \\
& =0.224 \mathrm{~L} \mathrm{CO}_2
\end{aligned}
$$
(C)
$12 \mathrm{~g}$ carbon on combustion gives
$$
=22.4 \mathrm{~L} \mathrm{CO}_2
$$
$2.4 \mathrm{~g}$ carbon on combustion will give
$$
\begin{aligned}
& =\frac{22.4 \times 2.4}{12} \mathrm{LCO}_2 \\
& =2 \times 2.24 \mathrm{LCO}_2 \\
& =4.48 \mathrm{LCO}_2
\end{aligned}
$$
(D)
$56 \mathrm{~g}$ carbon monoxide on combustion gives $=2 \times 22.4 \mathrm{~L} \mathrm{CO}_2$
$0.56 \mathrm{~g}$ carbon monoxide on combustion will give
$$
\begin{aligned}
& =\frac{2 \times 22.4 \times 0.56}{56} \mathrm{LCO}_2 \\
& =0.448 \mathrm{LCO}_2
\end{aligned}
$$
Hence, A-(iv), B-(i), C-(ii), D-(iii)
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.