Search any question & find its solution
Question:
Answered & Verified by Expert
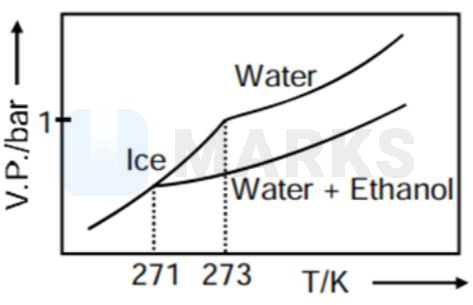
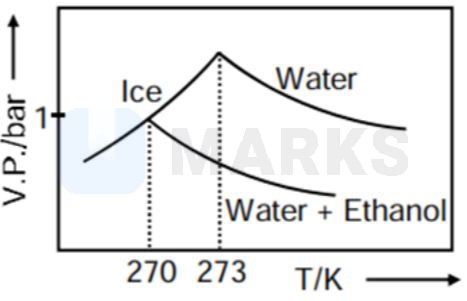
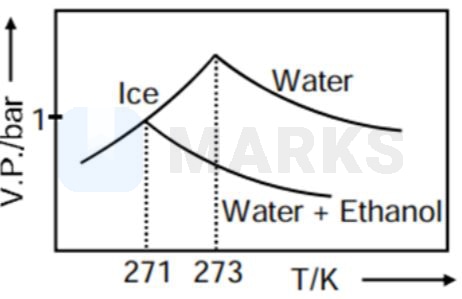
Pure water freezes at 273 K and 1 bar. The addition of 34.5 g of ethanol to 500 g of water changes the freezing point of the solution. Use the freezing point depression constant of water as 2 K kg . The figures shown below represent plots of vapour pressure (V.P.) versus temperature (T). [molecular weight of ethanol is 46 g ]
Among the following, the option representing change in the freezing point is
Options:
Among the following, the option representing change in the freezing point is
Solution:
1875 Upvotes
Verified Answer
The correct answer is:
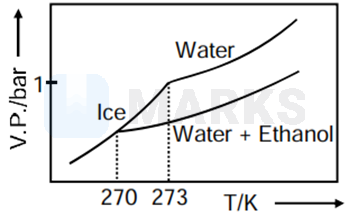

As when temperature increase, vapour pressure increases so C and D options get rejected.
So
So
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.