Search any question & find its solution
Question:
Answered & Verified by Expert
Rank the compounds given below in order of decreasing basicity
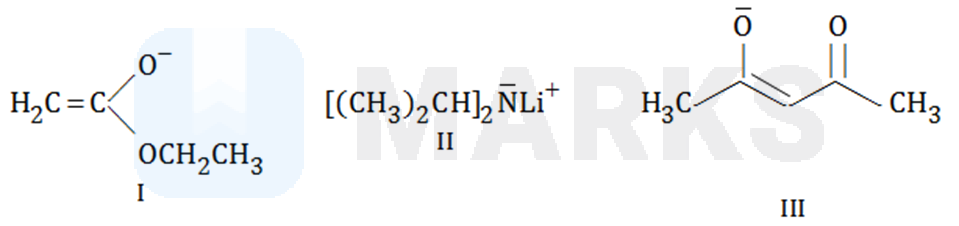
Options:
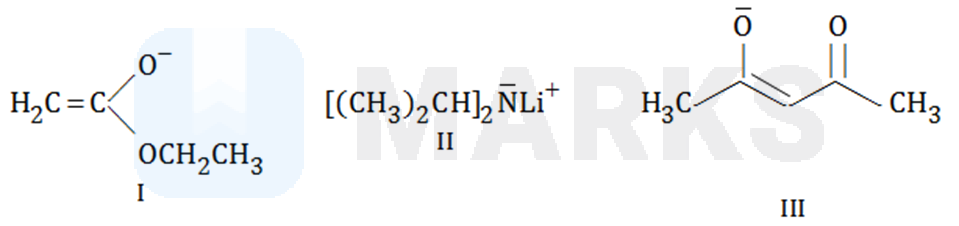
Solution:
2141 Upvotes
Verified Answer
The correct answer is:
II, I, III
Here the correct of basicity is
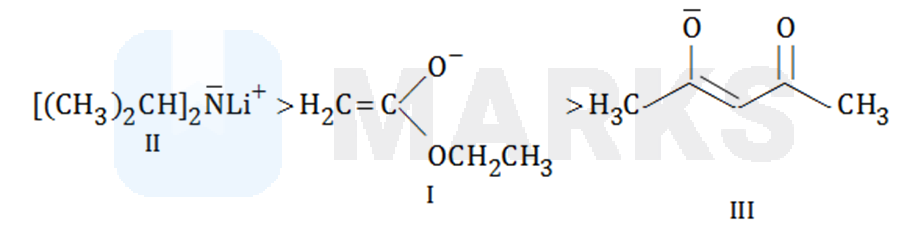
III is the weakest base since it is resonance stabilized and both resonance structures have negative charge on more electronegative (oxygen) atom.
I is weaker base than II since former has resonance stability.
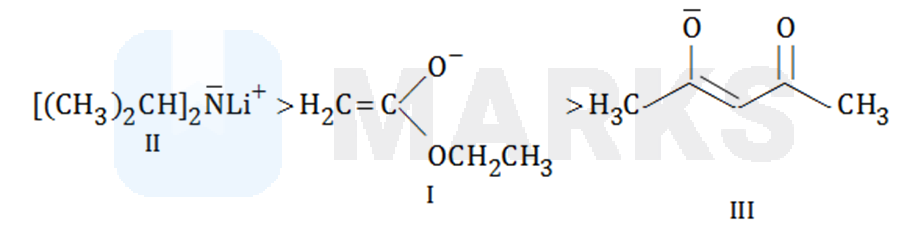
III is the weakest base since it is resonance stabilized and both resonance structures have negative charge on more electronegative (oxygen) atom.
I is weaker base than II since former has resonance stability.
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.