Search any question & find its solution
Question:
Answered & Verified by Expert
Reaction of $\mathrm{HBr}$ with propene in the presence of peroxide gives:
Options:
Solution:
2726 Upvotes
Verified Answer
The correct answer is:
$n$-propyl bromide
Reaction of $\mathrm{HBr}$ with propene in the presence of peroxide gives $n$-propyl bromide. This addition reaction is an example of anti-Markownikoffs addition reaction.
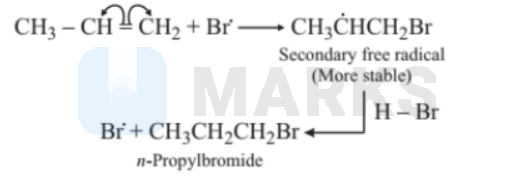
Related Theory
Step 1: Peroxide undergoes fission to give free radicals.
$$
\mathrm{R}-\mathrm{O}-\mathrm{O}-\mathrm{R} \longrightarrow 2 \mathrm{R}-\dot{\mathrm{O}}
$$
Step 2: $\mathrm{HBr}$ combines with free radical to form bromine free radical.
$$
R-\dot{O}+\mathrm{HBr} \longrightarrow \mathrm{R}-\mathrm{OH}+\dot{\mathrm{B}}
$$
Step 3: $\dot{B}$ attacks the double bond of the alkene to form a more stable free radical.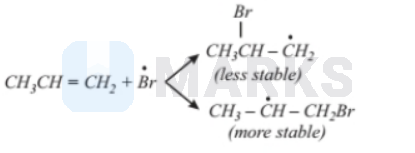
Step 4: More stable free radical attacks the $\mathrm{HBr}$.
$$
\begin{aligned}
\mathrm{CH}_3 \dot{\mathrm{C}} \mathrm{CHH}_2 \mathrm{Br}+\mathrm{HBr} \longrightarrow & \mathrm{CH}_3 \mathrm{CH}_2 \mathrm{CH}_2 \mathrm{Br}+\dot{\mathrm{B}} \\
& \text { n-propylbromide }
\end{aligned}
$$
Step 5: $\dot{B} r+\dot{B} r \longrightarrow B r_2$
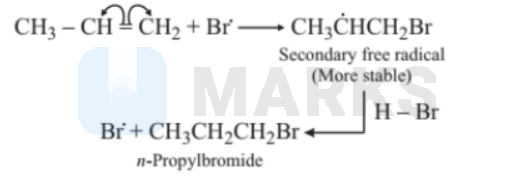
Related Theory
Step 1: Peroxide undergoes fission to give free radicals.
$$
\mathrm{R}-\mathrm{O}-\mathrm{O}-\mathrm{R} \longrightarrow 2 \mathrm{R}-\dot{\mathrm{O}}
$$
Step 2: $\mathrm{HBr}$ combines with free radical to form bromine free radical.
$$
R-\dot{O}+\mathrm{HBr} \longrightarrow \mathrm{R}-\mathrm{OH}+\dot{\mathrm{B}}
$$
Step 3: $\dot{B}$ attacks the double bond of the alkene to form a more stable free radical.

Step 4: More stable free radical attacks the $\mathrm{HBr}$.
$$
\begin{aligned}
\mathrm{CH}_3 \dot{\mathrm{C}} \mathrm{CHH}_2 \mathrm{Br}+\mathrm{HBr} \longrightarrow & \mathrm{CH}_3 \mathrm{CH}_2 \mathrm{CH}_2 \mathrm{Br}+\dot{\mathrm{B}} \\
& \text { n-propylbromide }
\end{aligned}
$$
Step 5: $\dot{B} r+\dot{B} r \longrightarrow B r_2$
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.