Search any question & find its solution
Question:
Answered & Verified by Expert
Reversible expansion of an ideal gas under isothermal and adiabatic conditions are as shown in the figure.
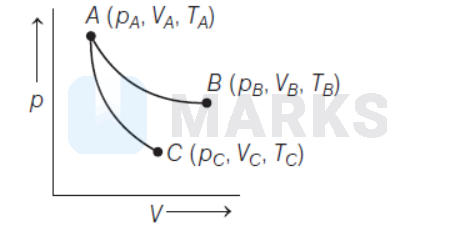
$A B \rightarrow$ Isothermal expansion
$A C \rightarrow$ Adiabatic expansion
Which of the following option is not correct?
Options:

$A B \rightarrow$ Isothermal expansion
$A C \rightarrow$ Adiabatic expansion
Which of the following option is not correct?
Solution:
2337 Upvotes
Verified Answer
The correct answer is:
$T_c>T_A$
From first law of thermodynamics,
$$
\Delta \mathrm{U}=\mathrm{q}+\mathrm{W}
$$
In adiabatic expansion, $q=0$
$$
\therefore \quad \Delta \mathrm{U}=\mathrm{W}
$$
During expansion of a gas $w$ is negative i.e $w < 0$ or $\Delta \mathrm{U} < 0$.
We know that, $\Delta \mathrm{U}=\mathrm{nC}_{\mathrm{V}} \Delta \mathrm{T}$
$$
\begin{array}{lclc}
\therefore & \mathrm{nC}_{\mathrm{V}} \Delta \mathrm{T} < 0 & \text { or } & \Delta \mathrm{T} < 0 \\
\therefore & \mathrm{T}_{\mathrm{C}}-\mathrm{T}_{\mathrm{A}} < 0 & \text { or } & \mathrm{T}_{\mathrm{C}} < \mathrm{T}_{\mathrm{A}}
\end{array}
$$
Thus, option (4) is incorrect while the remaining options are correct.
$$
\Delta \mathrm{U}=\mathrm{q}+\mathrm{W}
$$
In adiabatic expansion, $q=0$
$$
\therefore \quad \Delta \mathrm{U}=\mathrm{W}
$$
During expansion of a gas $w$ is negative i.e $w < 0$ or $\Delta \mathrm{U} < 0$.
We know that, $\Delta \mathrm{U}=\mathrm{nC}_{\mathrm{V}} \Delta \mathrm{T}$
$$
\begin{array}{lclc}
\therefore & \mathrm{nC}_{\mathrm{V}} \Delta \mathrm{T} < 0 & \text { or } & \Delta \mathrm{T} < 0 \\
\therefore & \mathrm{T}_{\mathrm{C}}-\mathrm{T}_{\mathrm{A}} < 0 & \text { or } & \mathrm{T}_{\mathrm{C}} < \mathrm{T}_{\mathrm{A}}
\end{array}
$$
Thus, option (4) is incorrect while the remaining options are correct.
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.