Search any question & find its solution
Question:
Answered & Verified by Expert
Show that in a cubic close packed structure, eight tetrahedral voids are present per unit cell.
Solution:
1749 Upvotes
Verified Answer
Cubic close packed structure contains one atom at each corner of an unit cell and one at face centre of each unit cell. Each unit cell consists of 8 small cubes.
Each small cube contains 4 atoms at its alternate corner when these atoms are joined to each other lead to creation of a tetrahedral void as shown in the figure below.
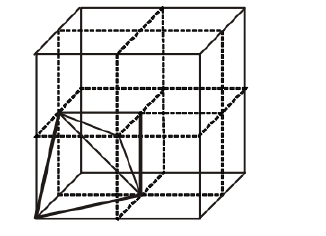
(a) Eight tetrahedral voids per unit cell of cep structure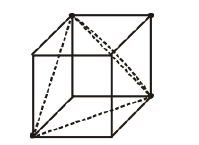
(b) One tetrahedral void showing the geometry
Since, there are total 8 smaller cubes present at one unit cell and each smaller cube has one tetrahedral void hence, total number of tetrahedral void present in each unit cell is equal to eight.
Each small cube contains 4 atoms at its alternate corner when these atoms are joined to each other lead to creation of a tetrahedral void as shown in the figure below.

(a) Eight tetrahedral voids per unit cell of cep structure

(b) One tetrahedral void showing the geometry
Since, there are total 8 smaller cubes present at one unit cell and each smaller cube has one tetrahedral void hence, total number of tetrahedral void present in each unit cell is equal to eight.
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.