Search any question & find its solution
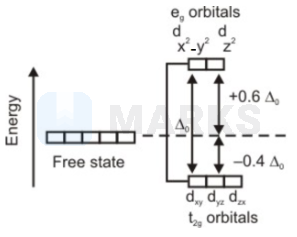 The above splitting of d-orbitals takes place in the formation of complexes. The above splitting of d-orbitals takes place in the formation of complexes.
The above splitting of d-orbitals takes place in the formation of complexes. The above splitting of d-orbitals takes place in the formation of complexes.In an octahedral coordination entity with six ligands surrounding the metal atom/ion, there will be repulsion between the electrons in metal d orbitals and the electrons (or negative charges) of the ligands.
Such a repulsion is more when the metal d orbital is directed towards the ligand than when it is away from the ligand. Thus, the and orbitals that point towards the axes along the direction of the ligand will experience more repulsion and will be raised in energy; and the orbitals which are directed between the axes will be lowered in energy relative to the average energy in the spherical crystal field.
Thus, the given diagram explains d orbital splitting in an octahedral crystal field.
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.