Search any question & find its solution
Question:
Answered & Verified by Expert
The compound has a total of ___ structural isomers.
Options:
Solution:
2955 Upvotes
Verified Answer
The correct answer is:
Compounds having the same molecular formula but different structures (manners in which atoms are linked) are classified as structural isomers. This includes chain isomers, position isomers, functional group isomers and metamerism.
For the given compound, , different structural isomers are possible, which are chain or position isomers of each other. The isomers are given below.
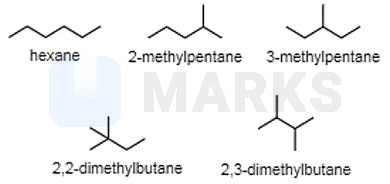
Therefore, the compound has a total of 5 structural isomers.
For the given compound, , different structural isomers are possible, which are chain or position isomers of each other. The isomers are given below.

Therefore, the compound has a total of 5 structural isomers.
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.