Search any question & find its solution
Question:
Answered & Verified by Expert
The correct statement regarding the basicity of aryl amines is:
Options:
Solution:
2820 Upvotes
Verified Answer
The correct answer is:
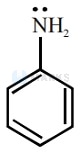
Aryl amines are generally less basic than alkyl amines because the nitrogen lone-pair electrons are delocalized by interaction with the aromatic ring electron system.
In Aryl amines lone pair is in conjugation with double bond so basic strength decreased
While in aliphatic amine no delocalisation of lone pair and +I effect of group so they have more basic strength.
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.