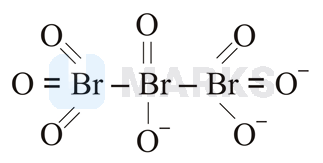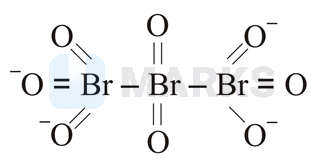Search any question & find its solution
Question:
Answered & Verified by Expert
The correct structure of tribromooctaoxide is:
Options:
Solution:
2381 Upvotes
Verified Answer
The correct answer is:
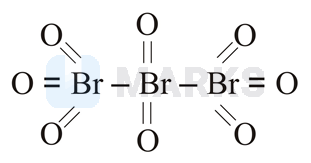
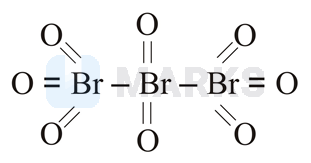
Tribromo octaoxide is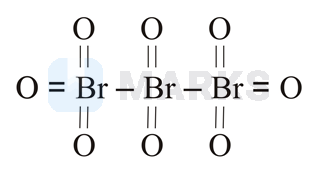
Lewis structures, also known as Lewis dot formulas, Lewis dot structures, electron dot structures, or Lewis electron dot structures, are diagrams that show the bonding between atoms of a molecule, as well as the lone pairs of electrons that may exist in the molecule.
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.