Search any question & find its solution
Question:
Answered & Verified by Expert
The following reaction descomposes At a degree centigrade the rate constant of the reaction is . The initial pressure of is If total pressure of gaseous mixture becomes then calculate the time of decomposition of .
Options:
Solution:
1939 Upvotes
Verified Answer
The correct answer is:
(i) The decomposition reaction is represented by
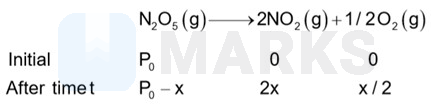
Total pressure
or
Pressure of after seconds,
Now applying first order rate law
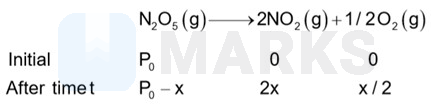
Total pressure
or
Pressure of after seconds,
Now applying first order rate law
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.