Search any question & find its solution
Question:
Answered & Verified by Expert
The following reaction method
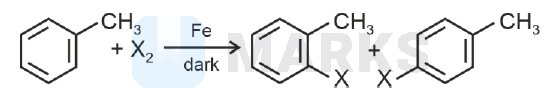
is not suitable for the preparation of the corresponding haloarene products, due to high reactivity of halogen, when X is :
Options:

is not suitable for the preparation of the corresponding haloarene products, due to high reactivity of halogen, when X is :
Solution:
2058 Upvotes
Verified Answer
The correct answer is:
F
- Aryl chlorides and bromides can easily be prepared by electrophilic substitution of arenes (toluene) with $\mathrm{Cl}_2$ and $\mathrm{Br}_2$ respectively in the presence of Lewis acid catalyst ( Fe in dark).
- Reaction with $\mathrm{I}_2$ is reversible and requires the presence of oxidising agent.
- Corresponding fluoroarene is not prepared by this method due to high reactivity of fluorine. Hence, ' X ' is $F$.
- Reaction with $\mathrm{I}_2$ is reversible and requires the presence of oxidising agent.
- Corresponding fluoroarene is not prepared by this method due to high reactivity of fluorine. Hence, ' X ' is $F$.
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.