Search any question & find its solution
Question:
Answered & Verified by Expert
The gas phase decomposition of dimethylether follows first order kinetics
The reaction is carried out in constant volume container at and has a half-life of . Initially only dimethylether is present at a pressure of . The total pressure of the system after is . The value of is
[Given ]
The reaction is carried out in constant volume container at and has a half-life of . Initially only dimethylether is present at a pressure of . The total pressure of the system after is . The value of is
[Given ]
Solution:
1445 Upvotes
Verified Answer
The correct answer is:
75
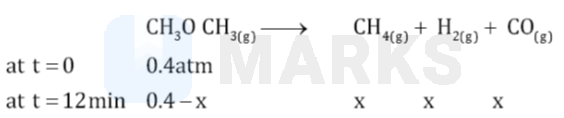
Now:
Total pressure
now:
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.