Search any question & find its solution
Question:
Answered & Verified by Expert
The hybridisation of each carbon in the following compound respectively is
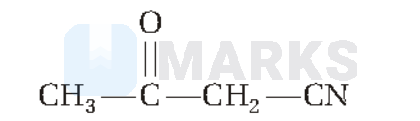
Options:
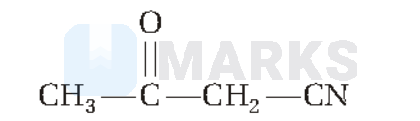
Solution:
1304 Upvotes
Verified Answer
The correct answer is:
$s p^3, s p^2, s p^3, s p$
In the compound,
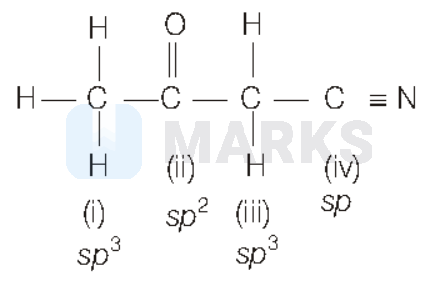
The $C$-atoms that is bonded to four different atoms involve four orbitals ( 1 of $s$ and 3 of $p$-orbitals) during
hybridisation. Hence, they have $s p^3$-hybridisation as in cases of (i) and (iii).
The $\mathrm{C}-$ atom bonded to three different atoms involves three orbitals ( 1 of $s$ and 2 of $p$-orbitals). Hence, the $s p^2$-hybridisation in case of (ii).
The $C$-atom bonded to two different atoms involves two orbitals ( 1 of $s$ and 1 of $p$-orbitals). Hence, the $s p$ - hybridisation in case of (iv).
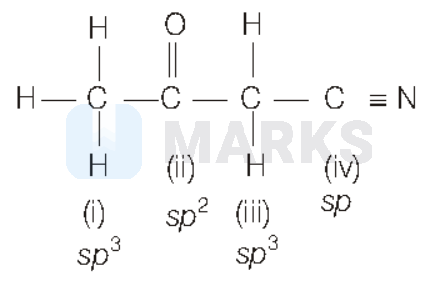
The $C$-atoms that is bonded to four different atoms involve four orbitals ( 1 of $s$ and 3 of $p$-orbitals) during
hybridisation. Hence, they have $s p^3$-hybridisation as in cases of (i) and (iii).
The $\mathrm{C}-$ atom bonded to three different atoms involves three orbitals ( 1 of $s$ and 2 of $p$-orbitals). Hence, the $s p^2$-hybridisation in case of (ii).
The $C$-atom bonded to two different atoms involves two orbitals ( 1 of $s$ and 1 of $p$-orbitals). Hence, the $s p$ - hybridisation in case of (iv).
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.