Search any question & find its solution
Question:
Answered & Verified by Expert
The hybridisations of and shown in the following compound
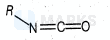
respectively, are
Solution:
2086 Upvotes
Verified Answer
The correct answer is:
Hybridisation is determined from the steric number (number of atoms bonded to the central atom + the number of lone pairs). Number of hybrid orbitals must be equal to the steric number.
From the Lewis structure.
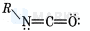
(i) Steric number of N-atom (2 bonded atoms +1 lone pair), Hybridisation (3 hybrid orbitals).
(ii) Steric number of C-atom (2 bonded atoms),
: Hybridisation ( 2 hybrid orbitals).
(iii) Steric number of 0 -atom (1 bonded atom +2 lone pair) Hybridisation hybrid orbitals).
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.