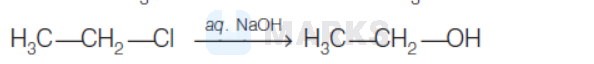Search any question & find its solution
Question:
Answered & Verified by Expert
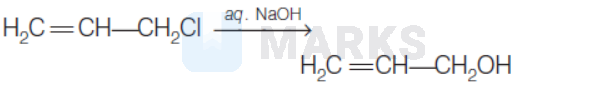
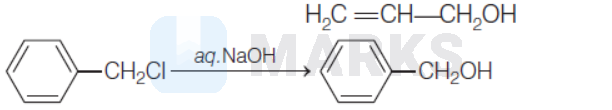
The hydrolysis reaction that takes place at the slowest rate, among the following is
Options:
Solution:
1406 Upvotes
Verified Answer
The correct answer is:
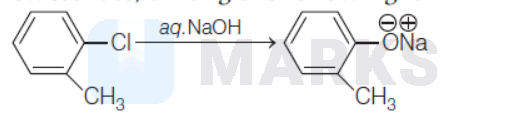

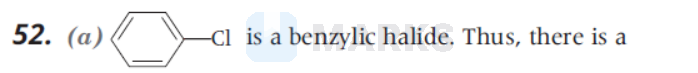
partial double bond character between $\mathrm{sp}^3$-hybridised $\mathrm{C}$ atom next to an aromatic ring and $\mathrm{Cl}$. It is most difficult to break this bond and hence it undergoes hydrolysis reaction with slowest rate.
Looking for more such questions to practice?
Download the MARKS App - The ultimate prep app for IIT JEE & NEET with chapter-wise PYQs, revision notes, formula sheets, custom tests & much more.